Advertisements
Advertisements
Question
Classify the following amine as primary, secondary or tertiary:
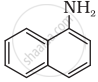
Options
Primary
Secondary
Tertiary
Advertisements
Solution
Primary
APPEARS IN
RELATED QUESTIONS
Classify the following amine as primary, secondary or tertiary:
(C2H5)2NH
Give reasons for the following:
CH3NH2 is more basis than C6H5NH2.
Choose the most correct option.
Carbylamine test is given by ____________.
Assertion: Acetamide on reaction with KOH and bromine gives acetic acid.
Reason: Bromine catalyses hydrolysis of acetamide.
When aniline reacts with acetic anhydride the product formed is ____________.
Which one of the following is most basic?
IUPAC name for the amine is:
\[\begin{array}{cc}
\phantom{.}\ce{CH3}\\
|\phantom{..}\\
\ce{CH3 - N - C - CH2 - CH3}\\
\phantom{.}|\phantom{.....}|\phantom{........}\\
\phantom{}\ce{CH3}\phantom{..}\ce{C2H5}\phantom{....}
\end{array}\]
Write a short note on the following.
Mustard oil reaction
How will you distinguish between primary secondary and tertiary aliphatic amines?
Arrange the following.
In decreasing order of basic strength in gas phase (C2H5)NH2, (C2H5)NH, (C2H5)3N and NH3.
Arrange the following.
In decreasing order of basic strength
![]()
How will you prepare propan-1-amine from 1-nitropropane?
How will you convert diethylamine into N, N-diethyl acetamide?
How will you convert diethylamine into N-nitrosodiethylamine?
The following amine can be classified as (C2H5)2NH:
\[\begin{array}{cc}
\ce{O}\phantom{.........}\\
||\phantom{.........}\\
\ce{H - \underset{(A)}{C} - NH - CH3}
\end{array}\]
and
\[\begin{array}{cc}
\ce{O}\\
||\\
\ce{CH3 - \underset{(B)}{C} - NH2}
\end{array}\]
are which type of isomers?
Define Amines.
Write a short note on the following
Ammonolysis
Write a short note on the following.
Ammonolysis
Write short note on Ammonolysis.
