Advertisements
Advertisements
Question
Give reasons for the following:
CH3NH2 is more basis than C6H5NH2.
Advertisements
Solution
Aromatic amines are far less basic than aliphatic amines. This can be explained as follows:
Resonance stabilisation is there in aniline. It can be regarded as a resonance hybrid of the following structures:
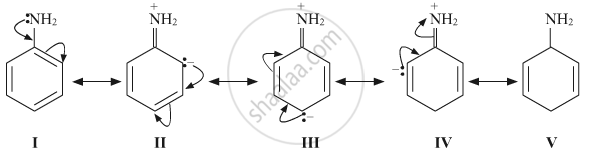
Hence, the lone pair of electrons on the nitrogen atom gets delocalised over benzene ring and thus is less available for protonation.
The electron density on the nitrogen atom is increased by electron-donating inductive effect of the alkyl groups. As a result, aliphatic amines are much stronger bases than aniline.
APPEARS IN
RELATED QUESTIONS
Classify the following amine as primary, secondary or tertiary:
(C2H5)2NH
Choose the most correct option.
Identify ‘B’ in the following reactions
\[\ce{CH3 - C ≡ N ->[Na/C2H5OH] A ->[NaNO2/dilHCl]B}\]
What are amines?
\[\ce{Aniline + benzoylchloride ->[NaOH] C6H5 - NH - COC6H5}\] this reaction is known as ____________.
When aniline reacts with acetic anhydride the product formed is ____________.
Among the following, which is the strongest base?
Write short notes on the following
Ammonolysis
Define Amines.
Write short note on Ammonolysis.
Write short note on the following:
Ammonolysis
