Advertisements
Advertisements
प्रश्न
Give reasons for the following:
CH3NH2 is more basis than C6H5NH2.
Advertisements
उत्तर
Aromatic amines are far less basic than aliphatic amines. This can be explained as follows:
Resonance stabilisation is there in aniline. It can be regarded as a resonance hybrid of the following structures:
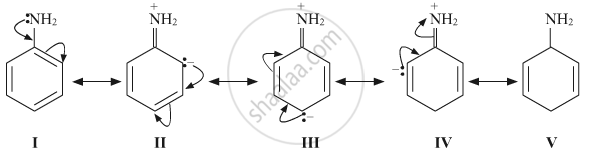
Hence, the lone pair of electrons on the nitrogen atom gets delocalised over benzene ring and thus is less available for protonation.
The electron density on the nitrogen atom is increased by electron-donating inductive effect of the alkyl groups. As a result, aliphatic amines are much stronger bases than aniline.
APPEARS IN
संबंधित प्रश्न
Classify the following amine as primary, secondary or tertiary:
(C2H5)2NH
Isobutylamine is an example of ______.
Choose the most correct option.
Carbylamine test is given by ____________.
What are amines?
How are amines classified?
IUPAC name for the amine is:
\[\begin{array}{cc}
\phantom{.}\ce{CH3}\\
|\phantom{..}\\
\ce{CH3 - N - C - CH2 - CH3}\\
\phantom{.}|\phantom{.....}|\phantom{........}\\
\phantom{}\ce{CH3}\phantom{..}\ce{C2H5}\phantom{....}
\end{array}\]
How will you prepare propan-1-amine from 1-nitropropane?
The following amine can be classified as (C2H5)2NH:
Arrange the following in the increasing order of their basic strength:
C2H5NH2, C6H5NH2, (C2H5)2NH
The reaction of NHO2 with 'A' gives quartering ammonium salt. A is which of the following?
