Advertisements
Advertisements
प्रश्न
Classify the following amine as primary, secondary or tertiary:
(C2H5)2NH
विकल्प
Primary
Secondary
Tertiary
Advertisements
उत्तर
Secondary
APPEARS IN
संबंधित प्रश्न
What are amines?
Which of the following amines is most basic in nature in aqueous phase?
\[\ce{Aniline + benzoylchloride ->[NaOH] C6H5 - NH - COC6H5}\] this reaction is known as ____________.
The product formed by the reaction an aldehyde with a primary amine ____________.
The order of basic strength for methyl substituted amines in aqueous solution is ____________.
Which of the following amines does not undergo acetylation?
Write a short note on the following:
Gabriel phthalimide synthesis
Write a short note on the following.
Mustard oil reaction
Account for the following.
Although amino group is o- and p-directing in aromatic electrophilic substitution reactions, aniline on nitration gives a substantial amount of m-nitroaniline.
How will you prepare propan-1-amine from butane nitrile?
How will you prepare propan-1-amine from 1-nitropropane?
Arrange the following in the increasing order of their basic strength:
C2H5NH2, C6H5NH2, (C2H5)2NH
Classify the following amine as primary, secondary or tertiary:
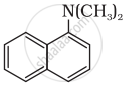
Which among the following is the strongest Bronsted base?
Among the following, which is the strongest base?
Write short note on the following:
Ammonolysis
Write a short note on the following.
Ammonolysis
Write a short note on the following.
Ammonolysis
Write a short note on the following.
Ammonolysis
