Advertisements
Advertisements
प्रश्न
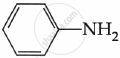
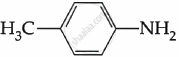
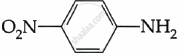
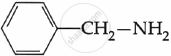
Among the following, which is the strongest base?
विकल्प
Advertisements
उत्तर
Explanation:
The electron density on the nitrogen atom increases due to the presence of the CH3 group.
APPEARS IN
संबंधित प्रश्न
Classify the following amine as primary, secondary or tertiary:
(C2H5)2NH
Choose the most correct option.
Identify ‘B’ in the following reactions
\[\ce{CH3 - C ≡ N ->[Na/C2H5OH] A ->[NaNO2/dilHCl]B}\]
How are amines classified?
Account for the following.
Although amino group is o- and p-directing in aromatic electrophilic substitution reactions, aniline on nitration gives a substantial amount of m-nitroaniline.
Identify A, B and C.
\[\ce{CH3 - NO2 ->[LiAlH4] A ->[2CH3CH2Br] B ->[H2SO4] C}\]
Arrange the following in the increasing order of their basic strength:
C2H5NH2, C6H5NH2, (C2H5)2NH
The amine formed from an amide by mean of bromine and alkali has how many number of carbon atoms?
Write short notes on the following
Ammonolysis
Write short note on the following.
Ammonolysis
Write short note on the following:
Ammonolysis