Advertisements
Advertisements
प्रश्न
Identify A, B and C.
\[\ce{CH3 - NO2 ->[LiAlH4] A ->[2CH3CH2Br] B ->[H2SO4] C}\]
Advertisements
उत्तर
\[\begin{array}{cc}
\ce{\underset{(Nitro methane)}{CH3NO2} ->[LiAlH4][6(H)/-2H2O] \underset{\underset{(A)}{(Methylamine)}}{CH3NH2} ->[2CH3CH2Br][-2HBr] \underset{\underset{(B)}{(N, N-diethyl methylamine)}}{(CH3CH2)2NCH3} ->[H2SO4] [(CH3CH2)2NH^+] HSO^-_4}\\
\phantom{..........................................................................}|\\
\phantom{.............................................................................}\ce{\underset{\underset{(C)}{(Quarternary salt)}}{CH3}}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
Choose the most correct option.
The hybridisation of nitrogen in primary amine is ____________.
Assertion: Acetamide on reaction with KOH and bromine gives acetic acid.
Reason: Bromine catalyses hydrolysis of acetamide.
\[\ce{Aniline + benzoylchloride ->[NaOH] C6H5 - NH - COC6H5}\] this reaction is known as ____________.
Account for the following.
pKb of aniline is more than that of methylamine.
The following amine can be classified as:

The amine formed from an amide by mean of bromine and alkali has how many number of carbon atoms?
Classify the following amine as primary, secondary or tertiary:
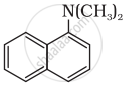
Write short note on the following.
Ammonolysis
Define Amines.
Write short note on Ammonolysis.
