Advertisements
Advertisements
प्रश्न
Classify the following amine as primary, secondary or tertiary:
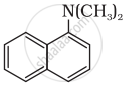
विकल्प
Primary
Secondary
Tertiary
Advertisements
उत्तर
Tertiary
APPEARS IN
संबंधित प्रश्न
Give reasons for the following:
CH3NH2 is more basis than C6H5NH2.
Choose the most correct option.
The hybridisation of nitrogen in primary amine is ____________.
Choose the most correct option.
Which type of amine does produce N2 when treated with HNO2?
What are amines?
Write the number of moles of ethanoyl chloride required for complete acylation of N, N-dimethylaniline.
A tertiary amine is an organic compound ____________.
Which of the following amines is most basic in nature in aqueous phase?
The order of basic strength for methyl substituted amines in aqueous solution is ____________.
IUPAC name for the amine is:
\[\begin{array}{cc}
\phantom{.}\ce{CH3}\\
|\phantom{..}\\
\ce{CH3 - N - C - CH2 - CH3}\\
\phantom{.}|\phantom{.....}|\phantom{........}\\
\phantom{}\ce{CH3}\phantom{..}\ce{C2H5}\phantom{....}
\end{array}\]
Write a short note on the following:
Gabriel phthalimide synthesis
Account for the following.
Gabriel phthalimide synthesis is preferred for synthesising primary amines.
Arrange the following:
In increasing order of solubility in water:
C6H5NH2, (C2H5)2NH, C2H5NH2
Arrange the following.
In decreasing order of basic strength in gas phase (C2H5)NH2, (C2H5)NH, (C2H5)3N and NH3.
The following amine can be classified as:

Complete the following reaction.
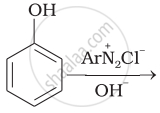
\[\begin{array}{cc}
\ce{O}\phantom{.........}\\
||\phantom{.........}\\
\ce{H - \underset{(A)}{C} - NH - CH3}
\end{array}\]
and
\[\begin{array}{cc}
\ce{O}\\
||\\
\ce{CH3 - \underset{(B)}{C} - NH2}
\end{array}\]
are which type of isomers?
Classify the following amine as primary, secondary or tertiary:
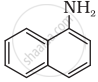
Write short note on Ammonolysis.
Write a short note on the following.
Ammonolysis
