Advertisements
Advertisements
प्रश्न
Classify the following amine as primary, secondary or tertiary:
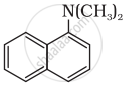
पर्याय
Primary
Secondary
Tertiary
Advertisements
उत्तर
Tertiary
APPEARS IN
संबंधित प्रश्न
Give reasons for the following:
CH3NH2 is more basis than C6H5NH2.
Choose the most correct option.
Carbylamine test is given by ____________.
What is the action of nitrous acid on the following compounds?
Isopropyl amine
Assertion: Acetamide on reaction with KOH and bromine gives acetic acid.
Reason: Bromine catalyses hydrolysis of acetamide.
The product formed by the reaction an aldehyde with a primary amine ____________.
Which of the following amines does not undergo acetylation?
Write a short note on the following.
Carbylamine reaction
Account for the following.
Ethylamine is soluble in water whereas aniline is not.
Arrange the following:
In increasing order of solubility in water:
C6H5NH2, (C2H5)2NH, C2H5NH2
Arrange the following.
Increasing order of basic strength C6H5NH2, C6H5N(CH3)2, (C2H5)2NH and CH3NH2.
How will you prepare propan-1-amine from propanamide?
The reaction of NHO2 with 'A' gives quartering ammonium salt. A is which of the following?
Which among the following is the strongest Bronsted base?
Among the following, which is the strongest base?
Arrange the increasing order of solubility in water.
\[\ce{C2H5Cl, C2H5NH2, C2H5OH}\]
Write short notes on the following
Ammonolysis
Write a short note on the following.
Ammonolysis
Write a short note on the following
Ammonolysis
Write a short note on the following.
Ammonolysis
