Advertisements
Advertisements
प्रश्न
Arrange the following.
Increasing order of basic strength C6H5NH2, C6H5N(CH3)2, (C2H5)2NH and CH3NH2.
Advertisements
उत्तर
Due to the delocalization of the lone pair of electrons of the N-atom over the benzene ring, all aromatic amines are less basic than alkylamines i.e., CH3NH2. The presence of electron-donating groups (– CH3) on the N-atom increases the basicity of substituted aniline with respect to C6H5NH2.
In (C6H5)NH2, the lone pair of electrons on the N-atom is delocalized over two benzene rings instead of one in C6H5NH2, therefore (C6H5)NH2 is much less basic than C6H5NH2. Combining all the three trends together, the basic strength of the four amines increasing in order.
(C6H5)NH2 < (C2H5)2NH < C6H5N(CH3)2 < CH3NH2
APPEARS IN
संबंधित प्रश्न
Choose the most correct option.
Identify ‘B’ in the following reactions
\[\ce{CH3 - C ≡ N ->[Na/C2H5OH] A ->[NaNO2/dilHCl]B}\]
\[\ce{C6H5NO2 ->[Fe/HCl] A ->[NaNO2/HCl][273 K] B ->[H2O][283 K] C}\] ‘C’ is:
IUPAC name for the amine is:
\[\begin{array}{cc}
\phantom{.}\ce{CH3}\\
|\phantom{..}\\
\ce{CH3 - N - C - CH2 - CH3}\\
\phantom{.}|\phantom{.....}|\phantom{........}\\
\phantom{}\ce{CH3}\phantom{..}\ce{C2H5}\phantom{....}
\end{array}\]
Account for the following.
Ethylamine is soluble in water whereas aniline is not.
Arrange the following:
In increasing order of solubility in water:
C6H5NH2, (C2H5)2NH, C2H5NH2
The reaction of NHO2 with 'A' gives quartering ammonium salt. A is which of the following?
Classify the following amine as primary, secondary or tertiary:
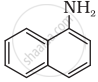
Classify the following amine as primary, secondary or tertiary:
(C2H5)2CHNH2
Write short note on the following.
Ammonolysis
Write a short note on the following.
Ammonolysis
