Advertisements
Advertisements
प्रश्न
Arrange the following.
In decreasing order of basic strength
![]()
Advertisements
उत्तर
Aliphatic amines are more basic than aromatic amines. Therefore CH3CH2NH2 and CH3NH2 are more basic. Among the ethylamine and methylamine. ethylamine was experienced more +I effect than methylamine and hence ethylamine is more basic than methylamine.
Nitro group has a powerful electron-withdrawing group and they have both – R effect as well as – I effect. As a result, all the nitro anilines are weaker bases than aniline. In P-nitro aniline
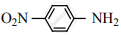
both – R effect and – I effect of the NO2 group decrease the basicity. Therefore decreasing order of basic strength is,
![]()
Ethylamine > Methylamine > Aniline > p-nitro aniline
APPEARS IN
संबंधित प्रश्न
Choose the most correct option.
The hybridisation of nitrogen in primary amine is ____________.
Choose the most correct option.
Carbylamine test is given by ____________.
IUPAC name for the amine is:
\[\begin{array}{cc}
\phantom{.}\ce{CH3}\\
|\phantom{..}\\
\ce{CH3 - N - C - CH2 - CH3}\\
\phantom{.}|\phantom{.....}|\phantom{........}\\
\phantom{}\ce{CH3}\phantom{..}\ce{C2H5}\phantom{....}
\end{array}\]
Account for the following.
Gabriel phthalimide synthesis is preferred for synthesising primary amines.
Arrange the following.
In decreasing order of the pKb values C2H5NH2, C6H5NHCH3, (C2H)2NH and CH3NH2.
How will you prepare propan-1-amine from butane nitrile?
Identify A, B and C.
\[\ce{CH3 - NO2 ->[LiAlH4] A ->[2CH3CH2Br] B ->[H2SO4] C}\]
How will you convert diethylamine into N, N-diethyl acetamide?
The reaction of NHO2 with 'A' gives quartering ammonium salt. A is which of the following?
Classify the following amine as primary, secondary or tertiary:
(C2H5)2CHNH2
