Advertisements
Advertisements
Question
Arrange the following.
In decreasing order of basic strength
![]()
Advertisements
Solution
Aliphatic amines are more basic than aromatic amines. Therefore CH3CH2NH2 and CH3NH2 are more basic. Among the ethylamine and methylamine. ethylamine was experienced more +I effect than methylamine and hence ethylamine is more basic than methylamine.
Nitro group has a powerful electron-withdrawing group and they have both – R effect as well as – I effect. As a result, all the nitro anilines are weaker bases than aniline. In P-nitro aniline
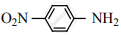
both – R effect and – I effect of the NO2 group decrease the basicity. Therefore decreasing order of basic strength is,
![]()
Ethylamine > Methylamine > Aniline > p-nitro aniline
APPEARS IN
RELATED QUESTIONS
Classify the following amine as primary, secondary or tertiary:
(C2H5)2NH
Choose the most correct option.
Which type of amine does produce N2 when treated with HNO2?
Choose the most correct option.
Identify ‘B’ in the following reactions
\[\ce{CH3 - C ≡ N ->[Na/C2H5OH] A ->[NaNO2/dilHCl]B}\]
What is the action of nitrous acid on the following compounds?
Isopropyl amine
Account for the following.
Gabriel phthalimide synthesis is preferred for synthesising primary amines.
Arrange the following.
In decreasing order of basic strength in gas phase (C2H5)NH2, (C2H5)NH, (C2H5)3N and NH3.
Classify the following amine as primary, secondary or tertiary:
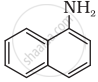
Classify the following amine as primary, secondary or tertiary:
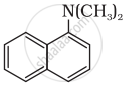
Write short note on the following:
Ammonolysis
Write a short note on the following.
Ammonolysis
