Advertisements
Advertisements
Question
Account for the following.
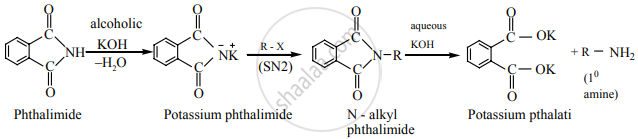
Gabriel phthalimide synthesis is preferred for synthesising primary amines.
Advertisements
Solution
Gabriel phthalimide reaction gives pure 1° amine without any contamination of 2° and 3° amines. Therefore it is preferred for synthesising primary amines.
APPEARS IN
RELATED QUESTIONS
Assertion: Acetamide on reaction with KOH and bromine gives acetic acid.
Reason: Bromine catalyses hydrolysis of acetamide.
\[\ce{Aniline + benzoylchloride ->[NaOH] C6H5 - NH - COC6H5}\] this reaction is known as ____________.
\[\ce{C6H5NO2 ->[Fe/HCl] A ->[NaNO2/HCl][273 K] B ->[H2O][283 K] C}\] ‘C’ is:
How will you distinguish between primary secondary and tertiary aliphatic amines?
Arrange the following:
In increasing order of solubility in water:
C6H5NH2, (C2H5)2NH, C2H5NH2
Arrange the following.
In decreasing order of basic strength
![]()
How will you prepare propan-1-amine from 1-nitropropane?
Among the following, which is the strongest base?
Name the distinguishing test for differentiating 1° amine from 2° and 3° amine.
Write short note on the following.
Ammonolysis
