Advertisements
Advertisements
Question
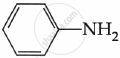
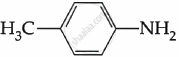
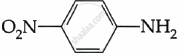
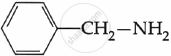
Among the following, which is the strongest base?
Options
Advertisements
Solution
Explanation:
The electron density on the nitrogen atom increases due to the presence of the CH3 group.
APPEARS IN
RELATED QUESTIONS
How are amines classified depending on the functional group? Give one example of each class of amines.
A tertiary amine is an organic compound ____________.
The product formed by the reaction an aldehyde with a primary amine ____________.
When aniline reacts with acetic anhydride the product formed is ____________.
Account for the following.
pKb of aniline is more than that of methylamine.
Arrange the following.
In decreasing order of the pKb values C2H5NH2, C6H5NHCH3, (C2H)2NH and CH3NH2.
Identify A, B and C.
\[\ce{CH3 - NO2 ->[LiAlH4] A ->[2CH3CH2Br] B ->[H2SO4] C}\]
Arrange the following in the increasing order of their basic strength:
C2H5NH2, C6H5NH2, (C2H5)2NH
Classify the following amine as primary, secondary or tertiary:
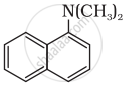
Write short note on the following:
Ammonolysis