Advertisements
Advertisements
Question
How are amines classified depending on the functional group? Give one example of each class of amines.
Advertisements
Solution
Classification of amines:
- Amines are classified as primary (1°), secondary (2°) and tertiary (3°) amines.
- Their structures are obtained in a simple way by replacing one, two, or three hydrogen atoms of NH3 molecule by alkyl/aryl groups.
- The functional group present in primary amines is referred to as an amino group (–NH2).
e.g. Ethylamine [C2H5 – NH2] - The functional group present in secondary amines is referred to as the imino group (>NH).
e.g. Dimethylamine [(CH3)2NH] - The functional group present in tertiary amines is tertiary nitrogen represented as:
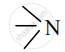
e.g. Trimethylamine [(CH3)3N]
APPEARS IN
RELATED QUESTIONS
Classify the following amine as primary, secondary or tertiary:
(C2H5)2NH
In the following

The compound ‘B’ is _______.
(A) Propan–1–amine
(B) Propan–2–amine
(C) Isopropylamine
(D) Dimethylamine
Isobutylamine is an example of ______.
Choose the most correct option.
Carbylamine test is given by ____________.
Choose the most correct option.
Identify ‘B’ in the following reactions
\[\ce{CH3 - C ≡ N ->[Na/C2H5OH] A ->[NaNO2/dilHCl]B}\]
How are amines classified?
Write the number of moles of ethanoyl chloride required for complete acylation of N, N-dimethylaniline.
What is the action of nitrous acid on the following compounds?
Isopropyl amine
Assertion: Acetamide on reaction with KOH and bromine gives acetic acid.
Reason: Bromine catalyses hydrolysis of acetamide.
\[\ce{Aniline + benzoylchloride ->[NaOH] C6H5 - NH - COC6H5}\] this reaction is known as ____________.
The product formed by the reaction an aldehyde with a primary amine ____________.
When aniline reacts with acetic anhydride the product formed is ____________.
The order of basic strength for methyl substituted amines in aqueous solution is ____________.
\[\ce{C6H5NO2 ->[Fe/HCl] A ->[NaNO2/HCl][273 K] B ->[H2O][283 K] C}\] ‘C’ is:
Which one of the following is most basic?
IUPAC name for the amine is:
\[\begin{array}{cc}
\phantom{.}\ce{CH3}\\
|\phantom{..}\\
\ce{CH3 - N - C - CH2 - CH3}\\
\phantom{.}|\phantom{.....}|\phantom{........}\\
\phantom{}\ce{CH3}\phantom{..}\ce{C2H5}\phantom{....}
\end{array}\]
Write a short note on the following.
Ammonolysis
Write a short note on the following:
Gabriel phthalimide synthesis
Write a short note on the following.
Schotten-Baumann reaction
Write a short note on the following.
Carbylamine reaction
Write a short note on the following.
Mustard oil reaction
How will you distinguish between primary secondary and tertiary aliphatic amines?
Arrange the following:
In increasing order of solubility in water:
C6H5NH2, (C2H5)2NH, C2H5NH2
Arrange the following.
In decreasing order of the pKb values C2H5NH2, C6H5NHCH3, (C2H)2NH and CH3NH2.
Arrange the following.
In decreasing order of basic strength
![]()
How will you prepare propan-1-amine from butane nitrile?
How will you prepare propan-1-amine from propanamide?
How will you prepare propan-1-amine from 1-nitropropane?
(C2H5)2CHNH2. The following amine can be classified as:
The following amine can be classified as (C2H5)2NH:
The following amine can be classified as:

The main product is formed by treating an alkyl or benzyl halide with excess ammonia ____________.
Complete the following reaction.
The amine formed from an amide by mean of bromine and alkali has how many number of carbon atoms?
\[\begin{array}{cc}
\ce{O}\phantom{.........}\\
||\phantom{.........}\\
\ce{H - \underset{(A)}{C} - NH - CH3}
\end{array}\]
and
\[\begin{array}{cc}
\ce{O}\\
||\\
\ce{CH3 - \underset{(B)}{C} - NH2}
\end{array}\]
are which type of isomers?
Classify the following amine as primary, secondary or tertiary:
Classify the following amine as primary, secondary or tertiary:
(C2H5)2CHNH2
Assertion A: Aniline on nitration yields ortho, meta and para nitro derivatives of aniline.
Reason R: Nitrating mixture is a storng acidic mixture.
In the light of the above statements, choose the correct answer from the options given below:
Among the following, which is the strongest base?
Arrange the increasing order of solubility in water.
\[\ce{C2H5Cl, C2H5NH2, C2H5OH}\]
Write short note on the following:
Ammonolysis
Write short notes on the following
Ammonolysis
Write short note on the following.
Ammonolysis
Define Amines.
Write a short note on the following.
Ammonolysis
Account for the following:
Aniline does not undergo Friedel-Crafts reaction.
Write short note on the following.
Ammonolysis
Write short note on the following:
Ammonolysis
Write a short note on the following.
Ammonolysis
