Advertisements
Advertisements
Question
Write a short note on the following:
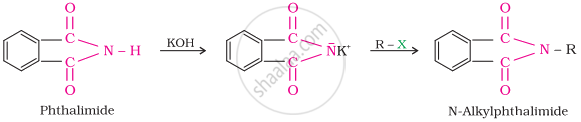
Gabriel phthalimide synthesis
Advertisements
Solution
Gabriel synthesis is used for the preparation of primary amines. Phthalimide, on treatment with ethanolic potassium hydroxide, forms potassium salt of phthalimide, which on heating with alkyl halide followed by alkaline hydrolysis, produces the corresponding primary amine. Aromatic primary amines cannot be prepared by this method because aryl halides do not undergo nucleophilic substitution with the anion formed by phthalimide.

RELATED QUESTIONS
Isobutylamine is an example of ______.
Choose the most correct option.
Which type of amine does produce N2 when treated with HNO2?
Choose the most correct option.
Carbylamine test is given by ____________.
Assertion: Acetamide on reaction with KOH and bromine gives acetic acid.
Reason: Bromine catalyses hydrolysis of acetamide.
When aniline reacts with acetic anhydride the product formed is ____________.
The order of basic strength for methyl substituted amines in aqueous solution is ____________.
Which one of the following is most basic?
Write a short note on the following.
Carbylamine reaction
Account for the following.
Gabriel phthalimide synthesis is preferred for synthesising primary amines.
Arrange the following.
In decreasing order of the pKb values C2H5NH2, C6H5NHCH3, (C2H)2NH and CH3NH2.
Arrange the following.
In decreasing order of basic strength
![]()
How will you prepare propan-1-amine from propanamide?
How will you convert diethylamine into N, N-diethyl acetamide?
(C2H5)2CHNH2. The following amine can be classified as:
The main product is formed by treating an alkyl or benzyl halide with excess ammonia ____________.
Classify the following amine as primary, secondary or tertiary:
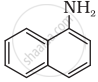
Classify the following amine as primary, secondary or tertiary:
(C2H5)2CHNH2
Among the following, which is the strongest base?
Write short notes on the following
Ammonolysis
Name the distinguishing test for differentiating 1° amine from 2° and 3° amine.
Write a short note on the following.
Ammonolysis
Write short note on the following:
Ammonolysis
Write a short note on the following.
Ammonolysis
