Advertisements
Advertisements
Question
How will you convert diethylamine into N, N-diethyl acetamide?
Advertisements
Solution
Conversion of diethylamine into N, N-diethyl acetamide:
Diethylamine reacts with acetyl chloride in the presence of pyridine to form N, N-diethyl acetamide.
\[\ce{\underset{(Diethylamine)}{(C2H5)2NH} + \underset{(Acetyl chloride)}{CH3COCl} ->[Pyridine] \underset{(N, N-diethyl acetamide)}{(C2H5)2N - CO - CH3} + HCl}\]
APPEARS IN
RELATED QUESTIONS
In the following

The compound ‘B’ is _______.
(A) Propan–1–amine
(B) Propan–2–amine
(C) Isopropylamine
(D) Dimethylamine
Isobutylamine is an example of ______.
What is the action of nitrous acid on the following compounds?
Isopropyl amine
Write a short note on the following.
Ammonolysis
How will you prepare propan-1-amine from propanamide?
The amine formed from an amide by mean of bromine and alkali has how many number of carbon atoms?
Classify the following amine as primary, secondary or tertiary:
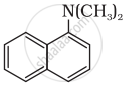
Assertion A: Aniline on nitration yields ortho, meta and para nitro derivatives of aniline.
Reason R: Nitrating mixture is a storng acidic mixture.
In the light of the above statements, choose the correct answer from the options given below:
Define Amines.
Write short note on Ammonolysis.
