Advertisements
Advertisements
Question
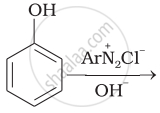
In the following

The compound ‘B’ is _______.
(A) Propan–1–amine
(B) Propan–2–amine
(C) Isopropylamine
(D) Dimethylamine
Advertisements
Solution
(A) Propan–1–amine
APPEARS IN
RELATED QUESTIONS
Choose the most correct option.
The hybridisation of nitrogen in primary amine is ____________.
Isobutylamine is an example of ______.
What are amines?
How are amines classified?
What is the action of nitrous acid on the following compounds?
Isopropyl amine
Which of the following amines is most basic in nature in aqueous phase?
Assertion: Acetamide on reaction with KOH and bromine gives acetic acid.
Reason: Bromine catalyses hydrolysis of acetamide.
\[\ce{Aniline + benzoylchloride ->[NaOH] C6H5 - NH - COC6H5}\] this reaction is known as ____________.
When aniline reacts with acetic anhydride the product formed is ____________.
\[\ce{C6H5NO2 ->[Fe/HCl] A ->[NaNO2/HCl][273 K] B ->[H2O][283 K] C}\] ‘C’ is:
Which of the following amines does not undergo acetylation?
Which one of the following is most basic?
IUPAC name for the amine is:
\[\begin{array}{cc}
\phantom{.}\ce{CH3}\\
|\phantom{..}\\
\ce{CH3 - N - C - CH2 - CH3}\\
\phantom{.}|\phantom{.....}|\phantom{........}\\
\phantom{}\ce{CH3}\phantom{..}\ce{C2H5}\phantom{....}
\end{array}\]
How will you convert nitrobenzene into m-nitro aniline?
Write a short note on the following.
Ammonolysis
Write a short note on the following:
Gabriel phthalimide synthesis
Write a short note on the following.
Schotten-Baumann reaction
Write a short note on the following.
Carbylamine reaction
How will you distinguish between primary secondary and tertiary aliphatic amines?
Account for the following.
pKb of aniline is more than that of methylamine.
Account for the following.
Ethylamine is soluble in water whereas aniline is not.
Arrange the following:
In increasing order of solubility in water:
C6H5NH2, (C2H5)2NH, C2H5NH2
Arrange the following.
In decreasing order of the pKb values C2H5NH2, C6H5NHCH3, (C2H)2NH and CH3NH2.
How will you prepare propan-1-amine from butane nitrile?
How will you prepare propan-1-amine from 1-nitropropane?
How will you convert diethylamine into N, N-diethyl acetamide?
How will you convert diethylamine into N-nitrosodiethylamine?
(C2H5)2CHNH2. The following amine can be classified as:
The following amine can be classified as:

The main product is formed by treating an alkyl or benzyl halide with excess ammonia ____________.
Complete the following reaction.
The amine formed from an amide by mean of bromine and alkali has how many number of carbon atoms?
\[\begin{array}{cc}
\ce{O}\phantom{.........}\\
||\phantom{.........}\\
\ce{H - \underset{(A)}{C} - NH - CH3}
\end{array}\]
and
\[\begin{array}{cc}
\ce{O}\\
||\\
\ce{CH3 - \underset{(B)}{C} - NH2}
\end{array}\]
are which type of isomers?
Classify the following amine as primary, secondary or tertiary:
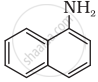
Classify the following amine as primary, secondary or tertiary:
(C2H5)2CHNH2
Among the following, which is the strongest base?
Write short note on the following:
Ammonolysis
Write short note on the following.
Ammonolysis
Define Amines.
Write a short note on the following
Ammonolysis
Write a short note on the following.
Ammonolysis
Write short note on the following.
Ammonolysis
Write short note on the following:
Ammonolysis
