Advertisements
Advertisements
प्रश्न
In the following

The compound ‘B’ is _______.
(A) Propan–1–amine
(B) Propan–2–amine
(C) Isopropylamine
(D) Dimethylamine
Advertisements
उत्तर
(A) Propan–1–amine
APPEARS IN
संबंधित प्रश्न
Classify the following amine as primary, secondary or tertiary:
(C2H5)2NH
Choose the most correct option.
Which type of amine does produce N2 when treated with HNO2?
Choose the most correct option.
Identify ‘B’ in the following reactions
\[\ce{CH3 - C ≡ N ->[Na/C2H5OH] A ->[NaNO2/dilHCl]B}\]
What are amines?
How are amines classified depending on the functional group? Give one example of each class of amines.
A tertiary amine is an organic compound ____________.
Which of the following amines is most basic in nature in aqueous phase?
The product formed by the reaction an aldehyde with a primary amine ____________.
When aniline reacts with acetic anhydride the product formed is ____________.
The order of basic strength for methyl substituted amines in aqueous solution is ____________.
\[\ce{C6H5NO2 ->[Fe/HCl] A ->[NaNO2/HCl][273 K] B ->[H2O][283 K] C}\] ‘C’ is:
Which one of the following is most basic?
IUPAC name for the amine is:
\[\begin{array}{cc}
\phantom{.}\ce{CH3}\\
|\phantom{..}\\
\ce{CH3 - N - C - CH2 - CH3}\\
\phantom{.}|\phantom{.....}|\phantom{........}\\
\phantom{}\ce{CH3}\phantom{..}\ce{C2H5}\phantom{....}
\end{array}\]
How will you convert nitrobenzene into m-nitro aniline?
Write a short note on the following:
Gabriel phthalimide synthesis
Write a short note on the following.
Schotten-Baumann reaction
Write a short note on the following.
Carbylamine reaction
Account for the following.
pKb of aniline is more than that of methylamine.
Account for the following.
Although amino group is o- and p-directing in aromatic electrophilic substitution reactions, aniline on nitration gives a substantial amount of m-nitroaniline.
Arrange the following:
In increasing order of solubility in water:
C6H5NH2, (C2H5)2NH, C2H5NH2
Arrange the following.
In decreasing order of basic strength in gas phase (C2H5)NH2, (C2H5)NH, (C2H5)3N and NH3.
Arrange the following.
In increasing order of boiling point C6H5OH, (CH3)2NH, C2H5NH2.
How will you prepare propan-1-amine from butane nitrile?
How will you prepare propan-1-amine from propanamide?
How will you convert diethylamine into N, N-diethyl acetamide?
How will you convert diethylamine into N-nitrosodiethylamine?
(C2H5)2CHNH2. The following amine can be classified as:
Arrange the following in the increasing order of their basic strength:
C2H5NH2, C6H5NH2, (C2H5)2NH
\[\begin{array}{cc}
\ce{O}\phantom{.........}\\
||\phantom{.........}\\
\ce{H - \underset{(A)}{C} - NH - CH3}
\end{array}\]
and
\[\begin{array}{cc}
\ce{O}\\
||\\
\ce{CH3 - \underset{(B)}{C} - NH2}
\end{array}\]
are which type of isomers?
Classify the following amine as primary, secondary or tertiary:
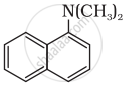
Classify the following amine as primary, secondary or tertiary:
(C2H5)2CHNH2
Which among the following is the strongest Bronsted base?
Assertion A: Aniline on nitration yields ortho, meta and para nitro derivatives of aniline.
Reason R: Nitrating mixture is a storng acidic mixture.
In the light of the above statements, choose the correct answer from the options given below:
Among the following, which is the strongest base?
Arrange the increasing order of solubility in water.
\[\ce{C2H5Cl, C2H5NH2, C2H5OH}\]
Write short note on the following:
Ammonolysis
Write short notes on the following
Ammonolysis
Name the distinguishing test for differentiating 1° amine from 2° and 3° amine.
Write a short note on the following
Ammonolysis
Write a short note on the following.
Ammonolysis
Account for the following:
Aniline does not undergo Friedel-Crafts reaction.
Write short note on Ammonolysis.
Write short note on the following:
Ammonolysis
Write a short note on the following.
Ammonolysis
Write a short note on the following.
Ammonolysis
