Advertisements
Advertisements
प्रश्न
Arrange the following.
In decreasing order of basic strength in gas phase (C2H5)NH2, (C2H5)NH, (C2H5)3N and NH3.
Advertisements
उत्तर
In the gas phase, solvent effects i.e., stabilization of the conjugate acids due to H-bonding, are absent. Therefore, in the gas phase, basic strength mainly depends upon the +I effect of the alkyl groups. Since the +I effect increases with the number of alkyl groups, therefore the basic strength of the amines decreases as the number of ethyl groups decreases from three in (C2H5)3N to two in (C2H5)2NH to one in C2H5NH2 and zero in NH3.
Basic strength in the gas phase decreases in the order is, (C2H5)3N > (C2H5)NH2 > (C2H5)NH > NH3.
APPEARS IN
संबंधित प्रश्न
Classify the following amine as primary, secondary or tertiary:
(C2H5)2NH
Which of the following amines does not undergo acetylation?
Write a short note on the following.
Schotten-Baumann reaction
Write a short note on the following.
Carbylamine reaction
Identify A, B and C.
\[\ce{CH3 - NO2 ->[LiAlH4] A ->[2CH3CH2Br] B ->[H2SO4] C}\]
How will you convert diethylamine into N, N-diethyl acetamide?
The main product is formed by treating an alkyl or benzyl halide with excess ammonia ____________.
Arrange the following in the increasing order of their basic strength:
C2H5NH2, C6H5NH2, (C2H5)2NH
Classify the following amine as primary, secondary or tertiary:
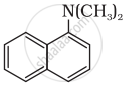
Write short note on Ammonolysis.
