Advertisements
Advertisements
प्रश्न
Arrange the following.
In increasing order of boiling point C6H5OH, (CH3)2NH, C2H5NH2.
Advertisements
उत्तर
Since the electronegativity of O is higher than that of N, therefore, alcohols form stronger H-O bonds than amines. In other words, the boiling points of alcohols are higher than those of amines of comparable molecular masses. Therefore the boiling point of C6H5OH (46) is higher than those of (CH3)2NH (45) and C2H5NH2 (45).
Further, the extent of H-bonding depends upon the number of H-atoms on the N-atom. Therefore 1° amine with two H-atoms on the N-atom has higher boiling points than 2° amines having only one H-atom. Therefore the boiling point of C2H5NH2 is higher than that of(CH3)2NH.
The increasing order of boiling point is, (CH3)2NH < C2H5NH2 < C6H5OH.
APPEARS IN
संबंधित प्रश्न
Classify the following amine as primary, secondary or tertiary:
(C2H5)2NH
Choose the most correct option.
The hybridisation of nitrogen in primary amine is ____________.
Choose the most correct option.
Identify ‘B’ in the following reactions
\[\ce{CH3 - C ≡ N ->[Na/C2H5OH] A ->[NaNO2/dilHCl]B}\]
Account for the following.
pKb of aniline is more than that of methylamine.
Arrange the following:
In increasing order of solubility in water:
C6H5NH2, (C2H5)2NH, C2H5NH2
Arrange the following.
In decreasing order of the pKb values C2H5NH2, C6H5NHCH3, (C2H)2NH and CH3NH2.
Classify the following amine as primary, secondary or tertiary:
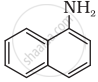
Name the distinguishing test for differentiating 1° amine from 2° and 3° amine.
Account for the following:
Aniline does not undergo Friedel-Crafts reaction.
Write a short note on the following.
Ammonolysis
