Advertisements
Advertisements
प्रश्न
Identify A, B and C.
\[\ce{CH3 - NO2 ->[LiAlH4] A ->[2CH3CH2Br] B ->[H2SO4] C}\]
Advertisements
उत्तर
\[\begin{array}{cc}
\ce{\underset{(Nitro methane)}{CH3NO2} ->[LiAlH4][6(H)/-2H2O] \underset{\underset{(A)}{(Methylamine)}}{CH3NH2} ->[2CH3CH2Br][-2HBr] \underset{\underset{(B)}{(N, N-diethyl methylamine)}}{(CH3CH2)2NCH3} ->[H2SO4] [(CH3CH2)2NH^+] HSO^-_4}\\
\phantom{..........................................................................}|\\
\phantom{.............................................................................}\ce{\underset{\underset{(C)}{(Quarternary salt)}}{CH3}}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
How are amines classified?
\[\ce{C6H5NO2 ->[Fe/HCl] A ->[NaNO2/HCl][273 K] B ->[H2O][283 K] C}\] ‘C’ is:
Which of the following amines does not undergo acetylation?
Write a short note on the following.
Mustard oil reaction
Account for the following.
Although amino group is o- and p-directing in aromatic electrophilic substitution reactions, aniline on nitration gives a substantial amount of m-nitroaniline.
How will you prepare propan-1-amine from propanamide?
The reaction of NHO2 with 'A' gives quartering ammonium salt. A is which of the following?
Classify the following amine as primary, secondary or tertiary:
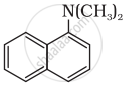
Name the distinguishing test for differentiating 1° amine from 2° and 3° amine.
Write a short note on the following.
Ammonolysis
