Advertisements
Advertisements
प्रश्न
Classify the following amine as primary, secondary or tertiary:
(C2H5)2NH
पर्याय
Primary
Secondary
Tertiary
Advertisements
उत्तर
Secondary
APPEARS IN
संबंधित प्रश्न
How are amines classified?
What is the action of nitrous acid on the following compounds?
Isopropyl amine
Assertion: Acetamide on reaction with KOH and bromine gives acetic acid.
Reason: Bromine catalyses hydrolysis of acetamide.
How will you convert nitrobenzene into m-nitro aniline?
Write a short note on the following.
Ammonolysis
Write a short note on the following.
Schotten-Baumann reaction
Write a short note on the following.
Mustard oil reaction
Account for the following.
Although amino group is o- and p-directing in aromatic electrophilic substitution reactions, aniline on nitration gives a substantial amount of m-nitroaniline.
Identify A, B and C.
\[\ce{CH3 - NO2 ->[LiAlH4] A ->[2CH3CH2Br] B ->[H2SO4] C}\]
How will you convert diethylamine into N, N-diethyl acetamide?
The main product is formed by treating an alkyl or benzyl halide with excess ammonia ____________.
Complete the following reaction.
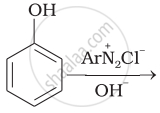
The reaction of NHO2 with 'A' gives quartering ammonium salt. A is which of the following?
Classify the following amine as primary, secondary or tertiary:
(C2H5)2CHNH2
Write short notes on the following
Ammonolysis
Define Amines.
Write a short note on the following.
Ammonolysis
Account for the following:
Aniline does not undergo Friedel-Crafts reaction.
Write a short note on the following.
Ammonolysis
Write a short note on the following.
Ammonolysis
