Advertisements
Advertisements
प्रश्न
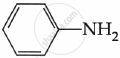
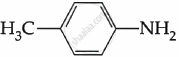
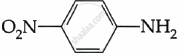
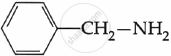
Among the following, which is the strongest base?
पर्याय
Advertisements
उत्तर
Explanation:
The electron density on the nitrogen atom increases due to the presence of the CH3 group.
APPEARS IN
संबंधित प्रश्न
Isobutylamine is an example of ______.
Choose the most correct option.
Which type of amine does produce N2 when treated with HNO2?
Which of the following amines is most basic in nature in aqueous phase?
Identify A, B and C.
\[\ce{CH3 - NO2 ->[LiAlH4] A ->[2CH3CH2Br] B ->[H2SO4] C}\]
How will you convert diethylamine into N, N-diethyl acetamide?
The following amine can be classified as:

The main product is formed by treating an alkyl or benzyl halide with excess ammonia ____________.
Assertion A: Aniline on nitration yields ortho, meta and para nitro derivatives of aniline.
Reason R: Nitrating mixture is a storng acidic mixture.
In the light of the above statements, choose the correct answer from the options given below:
Write short note on the following:
Ammonolysis
Name the distinguishing test for differentiating 1° amine from 2° and 3° amine.