Advertisements
Advertisements
Questions
Account for the following.
Ethylamine is soluble in water whereas aniline is not.
State reasons for the following:
Ethylamine is soluble in water whereas aniline is insoluble in water.
Advertisements
Solution 1
Ethylamine, when added to water, forms intermolecular H-bonds with water and therefore it is soluble in water. But aniline does not form an H-bond with water to a very large extent due to the presence of a large hydrophobic –C6H5 group. Hence, aniline is insoluble in water.
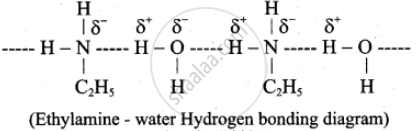
Solution 2
Ethylamine (C2H5NH2) exhibits solubility in water because the –NH group effectively forms hydrogen bonds with water molecules, due to the increased electron density on the nitrogen atom resulting from the +I inductive effect of the –C2H5 group. Also, the hydrocarbon part, –C2H5, is small and contributes much to hydrophobicity.
In Aniline (C6H5NH2), the –NH2 group is involved in resonance with the directly attached benzene ring, hence limiting its ability to form effective hydrogen bonds with water. Secondly, the benzene ring is substantial and generally hydrophobic. Hence, the net result is the insolubility of aniline in water.
RELATED QUESTIONS
Give reasons for the following:
CH3NH2 is more basis than C6H5NH2.
\[\ce{C6H5NO2 ->[Fe/HCl] A ->[NaNO2/HCl][273 K] B ->[H2O][283 K] C}\] ‘C’ is:
IUPAC name for the amine is:
\[\begin{array}{cc}
\phantom{.}\ce{CH3}\\
|\phantom{..}\\
\ce{CH3 - N - C - CH2 - CH3}\\
\phantom{.}|\phantom{.....}|\phantom{........}\\
\phantom{}\ce{CH3}\phantom{..}\ce{C2H5}\phantom{....}
\end{array}\]
How will you convert nitrobenzene into m-nitro aniline?
Write a short note on the following.
Mustard oil reaction
How will you distinguish between primary secondary and tertiary aliphatic amines?
Classify the following amine as primary, secondary or tertiary:
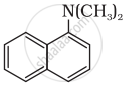
Name the distinguishing test for differentiating 1° amine from 2° and 3° amine.
Write a short note on the following
Ammonolysis
Write short note on the following:
Ammonolysis
