Advertisements
Advertisements
Question
Classify the following amine as primary, secondary or tertiary:
(C2H5)2CHNH2
Options
Primary
Secondary
Tertiary
Advertisements
Solution
Primary
APPEARS IN
RELATED QUESTIONS
Isobutylamine is an example of ______.
Choose the most correct option.
Identify ‘B’ in the following reactions
\[\ce{CH3 - C ≡ N ->[Na/C2H5OH] A ->[NaNO2/dilHCl]B}\]
What are amines?
How are amines classified?
How are amines classified depending on the functional group? Give one example of each class of amines.
IUPAC name for the amine is:
\[\begin{array}{cc}
\phantom{.}\ce{CH3}\\
|\phantom{..}\\
\ce{CH3 - N - C - CH2 - CH3}\\
\phantom{.}|\phantom{.....}|\phantom{........}\\
\phantom{}\ce{CH3}\phantom{..}\ce{C2H5}\phantom{....}
\end{array}\]
Account for the following.
pKb of aniline is more than that of methylamine.
Arrange the following.
In decreasing order of basic strength
![]()
How will you convert diethylamine into N, N-diethyl acetamide?
How will you convert diethylamine into N-nitrosodiethylamine?
Complete the following reaction.
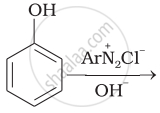
Classify the following amine as primary, secondary or tertiary:
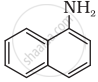
Which among the following is the strongest Bronsted base?
Write short note on the following:
Ammonolysis
Name the distinguishing test for differentiating 1° amine from 2° and 3° amine.
Define Amines.
Write short note on Ammonolysis.
