Advertisements
Advertisements
प्रश्न
Why Zn2+ ions are colourless while Ni2+ ions are green and Cu2+ ions are blue in colour?
Advertisements
उत्तर
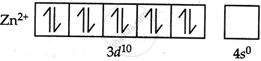
Zinc ions have a 3d orbital that is completely filled. Since the d orbitals of Zn2+ ions do not contain any unpaired electrons, they are colourless and do not absorb visible light because they do not show any electronic transitions that would produce visible colours.
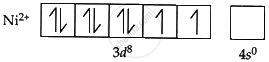
Two unpaired electrons make up the incomplete three-dimensional orbital of nickel ions (Ni2+). A complementary green color is seen for Ni2+ ions in solution when white light travels through it because some of the blue and green light is absorbed by the unpaired electrons in the 3d orbitals.
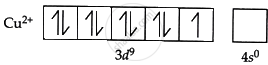
Also, copper ions (Cu2+) have one unpaired electron in an incomplete 3d orbital. The unpaired electrons in the 3d orbitals absorb the blue light when white light travels through a solution containing Cu2+ ions, giving the solution its complementary blue colour.