Advertisements
Advertisements
प्रश्न
A ketone A (C4H8O) which undergoes Iodoform reaction gives compound B on reduction. B on heating with conc.H2SO4 at 443 K gives a compound C which forms ozonide D. D on hydrolysis with Zn dust gives only E. Identify the compounds A to E. Write the Iodoform reaction with compound A.
Advertisements
उत्तर
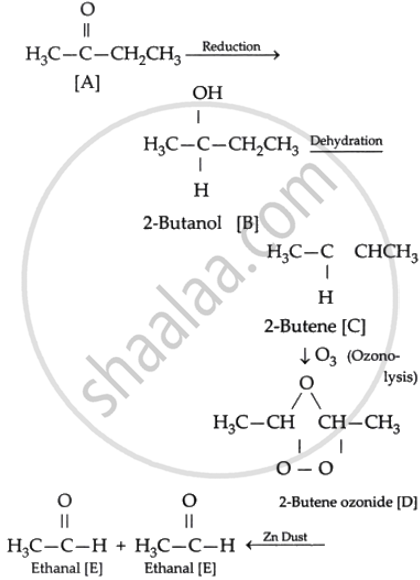
As the ketone molecule A exhibits a positive iodoform reaction, it is necessary for it to have a minimum of one free methyl group that is directly attached to the carbonyl carbon.
Ketone A must now be reduced to produce a secondary alcohol, which, when dehydrated, yields an alkene.
As an alkene undergoes ozonolysis to form an ozonide molecule, compound C should have one double bond.
As compound D produces only one product upon hydrolysis, the initial ketone should have two acetaldehyde molecules because it contains four C. It implies that compound C should have the 2 (CH3CH=) group.
Compound A, which satisfies all the above condition should be But-2-one.
The scheme for the reaction is as follows:
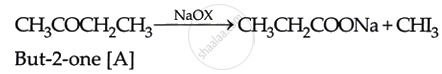
Iodoforrn reaction with compound A (But-2-one) is given as:
\[\ce{`CH3COCH2CH3 + 3I2 ++ 4NaOH -> CHI3 + C2H5COONa + 3NaI + 3H2O}\]
