- Carboxylic acids are carbon compounds with a –COOH group and have acidic nature.
- Types:
- Monocarboxylic acids have one –COOH group (e.g., formic acid, acetic acid)
- Dicarboxylic acids have two –COOH groups (e.g., oxalic acid) - IUPAC Naming: Replace the ‘e’ of the corresponding alkane with ‘oic acid’ (e.g., ethane → ethanoic acid); also called alkanoic acids.
Definitions [7]
Definition: Carbonyl Compound
Organic compounds containing carbon-oxygen double bond, i.e. \[\mathrm{>C=O}\] group, are known as carbonyl compounds.
Definition: Carboxylic Acid
An organic compound containing the carboxyl group (-COOH) is known as carboxylic acid. These compounds possess acidic properties.
Definition: Acyl halides
The organic compounds in which the –OH group of a carboxylic acid is replaced by a halogen atom are called acyl halides.
Definition: Esters
The organic compounds in which the –OH group of a carboxylic acid is replaced by an –OR group are called esters.
Definition: Acid anhydrides
The organic compounds formed by removal of one molecule of water from two molecules of carboxylic acid are called acid anhydrides.
Definition: Amides
The organic compounds in which the –OH group of a carboxylic acid is replaced by –NH₂ or substituted amino group are called amides.
Definition: Carbonyl group
The carbon–oxygen double bond (>C=O) functional group present in aldehydes, ketones and acids is called carbonyl group.
Formulae [5]
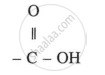
Formula: Carboxylic Acid
General formula: CnH2n+1COOH (or RCOOH)
Functional group:
Formula: Acyl halide
R–COX
Formula: Ester
R–COOR′
Formula: Acid anhydride
(R–CO)₂O
Formula: Amide
R–CONH₂
Chemica Equations [5]
Aldol Reaction
\[2CH_3CHO\xrightarrow{dil.NaOH}CH_3CH(OH)CH_2CHO\]
On heating:
→CH3CH = CHCHO + H2O
Cannizzaro Reaction
Esterification
\[RCOOH+R^{\prime}OH\xrightarrow{H^+}RCOOR^{\prime}+H_2O\]
Reduction
\[RCHO\xrightarrow{NaBH_4}RCH_2OH\]
\[RCOOH\xrightarrow{LiAlH_4}RCH_2OH\]
Decarboxylation
\[RCOONa+NaOH\xrightarrow{CaO}RH+Na_2CO_3\]
Key Points
Key Points: Concepts of Aldehydes, Ketones, and Carboxylic Acids
- Carbonyl group: The ≻C=O group (carbonyl carbon + carbonyl oxygen) — a key functional group in organic chemistry.
- Carbonyl compounds: Aldehydes and ketones, both containing ≻C=O as their functional group.
- Aldehydes: –CHO (formyl group); carbonyl C bonded to at least one H.
- Ketones: ≻C=O (ketonic carbonyl group); carbonyl C bonded to two alkyl/aryl groups (R=R′ or R≠R′).
- Carboxylic acids: –COOH (carboxyl group); –OH attached to ≻C=O makes them distinct from aldehydes/ketones.
Key Points: Preparation of Aldehydes and Ketones
- IUPAC rules: Longest chain with –CHO/–COOH is parent; –CHO/–COOH carbon gets number 1; two ≻C=O in ketones → suffix "dione".
- Preparation (common methods): Oxidation of 1° alcohols → aldehyde, 2° alcohols → ketone; dehydrogenation (Cu, 575 K); ozonolysis of alkenes; hydration of alkynes (dil. H₂SO₄ + HgSO₄, 333 K).
- Aldehyde-specific preparations: Rosenmund reduction (RCOCl, H₂/Pd–BaSO₄); Stephen reaction (RCN, SnCl₂·HCl, H₃O⁺); DIBAL-H on nitriles/esters; Etard reaction, Gatterman–Koch formylation for aromatic aldehydes.
- Ketone-specific preparations: Acyl chloride + R₂Cd; nitrile + R′MgX/dry ether, H₃O⁺; Friedel–Crafts acylation of benzene with RCOCl/anhyd. AlCl₃.
- Key reactions: HCN → cyanohydrin; NaHSO₃ → bisulphite adduct; R′OH/HCl → acetal/ketal; RMgX → 2°/3° alcohol; NH₂OH → oxime; NH₂NH₂ → hydrazone; Clemmensen (Zn-Hg/HCl) & Wolf–Kishner (NH₂NH₂/KOH) → alkane; K₂Cr₂O₇/H₂SO₄ → carboxylic acid.
Key Points: Physical Properties of Aldehydes and Ketones
- Preparation (aliphatic/aromatic): From 2° alcohol (K₂Cr₂O₇/H₂SO₄ or Cu/573 K), alkene (ozonolysis), alkyne (dil. H₂SO₄/HgSO₄, 333 K), acetyl chloride + R₂Cd, nitrile + R′MgX (dry ether, H₃O⁺).
- Aromatic ketones: Benzene + RCOCl / anhyd. AlCl₃ (Friedel–Crafts acylation).
- Addition reactions: HCN → cyanohydrin; NaHSO₃ → bisulphite adduct; R″OH/dry HCl → ketal; R′MgX then H⁺/H₂O → 3° alcohol.
- Condensation reactions: NH₂OH → ketoxime; NH₂NH₂ → hydrazone; C₆H₅NHNH₂ → phenylhydrazone.
- Reductions: Clemmensen (Zn–Hg/conc. HCl) and Wolf–Kishner (NH₂NH₂, KOH/ethylene glycol) → alkane (R–CH₂–R′).
Key Points: Carboxylic Acid
Key Points: Physical Properties of Carboxylic Acids
- Solubility: Decreases with an increase in the size of the hydrocarbon part.
- Miscibility: Lower carboxylic acids (up to 4 C atoms) are miscible with water due to H-bonding.
- Boiling point: Carboxylic acids have higher B.P. than ketones, aldehydes, and alcohols of comparable molecular mass due to intermolecular H-bonding.
- Order of B.P. (carboxylic acids & aldehydes): Valeric > Butyric > Propionic > Acetic > Formic acid; Hexanal > Pentanal > Butanal > Propanal.
- Order of B.P. (ketones): Hexan-2-one > Pentan-2-one > Butan-2-one > Propanone.
Key Points: Structure of Carbonyl Group
Statement:
The carbonyl carbon atom is sp² hybridised, forms a trigonal planar structure, and possesses a polar C=O bond.
Explanation:
1. Hybridisation:
- Carbonyl carbon is sp² hybridised.
- It forms:
- Three σ (sigma) bonds.
- One π (pi) bond with oxygen.
- The π-bond is formed by sideways overlap of p-orbitals.
2. Geometry:
- The carbonyl carbon and three attached atoms lie in the same plane.
- Bond angle ≈ 120°.
- Geometry is trigonal planar.
3. Polarity:
- Oxygen is more electronegative than carbon.
- Hence, electron density shifts towards oxygen.
- Carbon acquires partial positive charge (δ⁺).
- Oxygen acquires partial negative charge (δ⁻).
4. Resonance:
Two contributing structures:
R–C=O ↔ R–C⁺–O⁻
This explains:
- High dipole moment.
- Electrophilic nature of carbonyl carbon.
Conclusion:
The planar structure, polarity and resonance make the carbonyl carbon highly reactive towards nucleophiles.
Key Points: Reactivity of Aldehydes vs Ketones
Statement:
Aldehydes are more reactive than ketones towards nucleophilic addition reactions.
Reasons:
1. Steric Effect
- Aldehydes have: One alkyl group + one hydrogen.
- Ketones have: Two alkyl groups.
- More alkyl groups → more steric hindrance → less reactivity.
2. Electronic Effect (+I Effect)
- Alkyl groups show +I effect.
- They donate electron density to carbonyl carbon.
- This reduces partial positive charge on carbon.
Ketones (two alkyl groups) are less electrophilic.
Order of Reactivity:
Formaldehyde > Other aldehydes > Ketones
Example:
HCHO > CH₃CHO > CH₃COCH₃
Conclusion:
Due to lower steric hindrance and higher electrophilicity, aldehydes react faster than ketones.
Key Points: Reduction of Aldehydes and Ketones
Statement:
Carbonyl compounds undergo reduction to alcohols or hydrocarbons depending on reagents used.
Reduction to Alcohols
Reagents:
- NaBH₄
- LiAlH₄
- Catalytic hydrogenation (H₂/Ni)
Reactions:
Aldehyde → Primary alcohol
R–CHO → R–CH₂OH
Ketone → Secondary alcohol
R–CO–R′ → R–CHOH–R′
Reduction to Hydrocarbons
1. Clemmensen Reduction:
Zn(Hg)/HCl
R–CO–R′ → R–CH₂–R′
2. Wolff–Kishner Reduction:
NH₂NH₂/KOH
R–CO–R′ → R–CH₂–R′
Key Points:
- Carbonyl group converted to CH₂ group.
- Choice of reagent depends on acidic/basic conditions.
Conclusion:
Carbonyl compounds can be selectively reduced to alcohols or completely reduced to hydrocarbons.
Important Questions [139]
- Write the IUPAC name of the following complex: K2[PdCl4]
- Give the IUPAC name of the following compound. CH3 − CH = CH − CHO
- Write the Structure of 3-methyl Butanal
- Give Simple Chemical Tests to Distinguish Between the Following Pairs of Compounds: Butanal and Butan-2-one
- Using IUPAC norms write the formula for the following: Tetrahydroxidozincate (II)
- Using IUPAC norms, write the formula for the following: Pentaamminenitrito-N-Cobalt (III)
- Write the IUPAC name of the following complex: [Pt(NH3)6]Cl4
- Write the Structure of 2-methylbutanal.
- Write IUPAC name of the following compound:
- Complete the following: CHA3CN→2⋅HA2O1⋅AlH(i−Bu)A2A′A22′AA′→HA+HA2N−OHA′A22′BA′
- Arrange the following in the increasing order of their boiling points : C2H5OH, CH3 − CHO, CH3 − COOH
- Do the following conversions in not more than two steps: CH3COOH to CH3COCH3
- Do the following conversions in not more than two steps: CH2CH3 to COOH
- Write a Chemical Reaction to Show that the Open Structure of D-glucose Contains the Following : Aldehyde as a Carbonyl Group
- Draw the structure of the following compound. p-Methylbenzaldehyde
- Draw the structure of the following compound. 4-chloropentan-2-one
- Write the Iupac Name of the Following Compound:
- Complete the Following Reaction:
- Write the Structures of a and B in the Following Reactions
- Complete synthesis by giving missing starting material, reagent or product. →[(i) O3][(ii) Zn-H2O] 2 O
- Write the Reaction Involved in The Stephen Reduction
- How will you bring about the following conversion? Benzoyl chloride to benzaldehyde.
- Write the Chemical Equations to Illustrate the Following Name Reactions : Rosenmund Reduction
- Predict the reagent for carrying out the following transformations: Benzoyl chloride to Benzaldehyde
- Write the product in the following reaction
- Explain the following reactions: Stephan reaction
- Convert the following: Benzoic acid to Benzaldehyde.
- Write the Product in the Following Reaction
- Complete the synthesis by giving missing starting material, reagent or product. O || C
- Write the main product in the following reaction: 2CHA3COCl+(CHA3)A2Cd⟶
- Why is the boiling point of aldehydes and ketones lower than that of corresponding carboxylic acids?
- Give a chemical test to distinguish between ethanal acid and ethanoic acid.
- Arrange the following in the increasing order of their boiling points: CH3CHO, CH3COOH, CH3CH2OH
- Acetone, Acetaldehyde, Benzaldehyde, Acetophenone – Reactivity Towards Addition of Hcn.
- Arrange the following in the increasing order of their reactivity towards nucleophilic addition reaction: C6H5COCH3, CH3-CHO, CH3COCH3
- Predict the Products of the Following Reactions
- Draw the structure of the semicarbazone of ethanal.
- Write the Structures of the Main Products When Acetone (CH3 − CO − CH3) Reacts with the Following Reagent
- Write the products formed when CH3CHO reacts with the following reagents : HCN
- Write the products formed when CH3CHO reacts with the following reagents : H2N – OH
- Give a Simple Chemical Test to Distinguish Between
- Write the Main Product Formed When Propanal Reacts with the Following Reagents:
- Write the main product formed when propanal reacts with the following reagents: H2N- NH2 followed by heating with KOH in ethylene glycol.
- Arrange the following in the increasing order of their property indicated: Ethanal, Propanone, Propanal, Butanone (reactivity towards nucleophilic addition)
- What happens when propanone is treated with CH3MgBr and then hydrolysed?
- What happens when ethanal is treated with excess ethanol and acid?
- Which will undergo faster nucleophilic addition reaction? Acetaldehyde or Propanone
- Which of the following is most reactive in nucleophilic addition reactions?
- The carbon-oxygen double bond is polarised in aldehydes and ketones due to higher electronegativity of oxygen relative to carbon.
- Aldehydes and ketones react with hydroxylamine to form ______.
- Predict the Products of the Following Reactions : C H 3 C H 2 O H → C U / 573 K ?
- Predict the Products of the Following Reactions
- Etrh
- Write the chemical reaction involved in Wolff-Kishner reduction.
- Explain the following reactions: Clemmensen reaction
- Write Chemical Equations of the Following Reaction : Acetophenone is Treated with ("Zn"("Hg"))/"Conc.Hcl"^-
- Explain the following reaction: Wolff-Kishner reduction
- Write the main product in the following reaction: CHA3CHA2CHO→Zn(Hg)/Conc⋅HCl
- Write the structures of the main products when acetone (CH3 − CO − CH3) reacts with the following reagent
- How Will You Bring About the Following Conversions? Propanone to Propane
- Predict the Products of the Following Reactions : C H 3 − C H = C H 2 → I ) B 2 H 6 I I ) 3 H 2 O 2 / O H − ?
- What is the composition of Fehling's reagent?
- An organic compound 'A' with molecular formula C5H8O2 is reduced to n-pentane with hydrazine followed by heating with NaOH and glycol. 'A' forms a dioxime with hydroxylamine
- C 6 H 5 − O H → B R 2 ( a Q ) ?
- Distinguish Between: C6H5-COCH3 and C6H5-CHO
- Out of CH3CH2 – CO – CH2 – CH3 and CH3CH2 – CH2 – CO – CH3, which gives iodoform test?
- Give a simple chemical test to distinguish between the following pair of compounds: Benzaldehyde and Acetophenone
- An organic compound 'A' with the molecular formula C4H8O2 undergoes acid hydrolysis to form two compounds 'B' and 'C'.
- A and B are two functional isomers of compound C3H6O.On heating with NaOH and I2, isomer B forms yellow precipitate of iodoform whereas isomer A does not form any precipitate. Write the formulae of A and B.
- Give a simple chemical test to distinguish between the following pair of compounds: Propanal and Propanone
- Write chemical test to distinguish between the following compounds: Phenol and Benzoic acid
- The reagent that can be used to distinguish acetophenone and benzophenone is ______.
- Give a simple chemical test to distinguish between the following pair of compounds: Ethanal and Propanal
- Distinguish Between: CH3COOH and HCOOH
- Give a simple chemical test to distinguish between the following pair of compounds : CH3CH2CHO and CH3CH2COCH3
- Predict the Products of the Following Reactions
- Write Chemical Equations of the Following Reaction : Benzoyl Chloride is Hydrogenated in the Presence of "Pd"/("Baso"4^-)
- Convert the following: Acetaldehyde to But-2-enal
- Give Reasons Acetylation of Aniline Reduces Its Activation Effect.
- How will you convert ethanal into the following compound? But-2-enal
- Write the products formed when CH3CHO reacts with the following reagents: CH3CHO in the presence of dilute NaOH
- A Compound 'A' of Molecular Formula C2H3OCl Undergoes a Series of Reactions as Shown Below. Write the Structures of A, B, C and D in the Following Reactions
- How will you bring about the following conversion in not more than two steps? Ethanol to 3-Hydroxybutanal
- Assertion (A): The final product in Aldol condensation is always α, β-unsaturated carbonyl compound. Reason (R): α, β-unsaturated carbonyl compounds are stabilised due to conjugation.
- Which of the following does not give aldol condensation reaction?
- Why is Alpha (α) Hydrogen of Carbonyl Compounds Acidic in Nature?
- Which of the following compounds will undergo self-condensation in the presence of dilute NaOH solution?
- Why is the α-hydrogens of aldehydes and ketones are acidic in nature?
- Write a Chemical Equation for the Following Reaction : Propanone is Treated with Dilute Ba( Oh)2-.
- Predict the reagent for carrying out the following transformations: Ethanal to 3-hydroxy butanal
- Write the Product Formed When P-nitro Chlorobenzene is Heated with Aqueous Naoh at 443k Followed by Acidification?
- Complete the Following Reactions
- Write the Chemical Equations to Illustrate the Following Name Reactions: Aldol Condensation
- Write the equation involved in the following reaction: Etard reaction
- Which of the following does not give Cannizzaro reaction?
- Complete the Following Reaction:
- Write the Reactions Involved in the Following Reactions: Clemmensen Reduction
- Write the chemical equations to illustrate the following name reaction: Cannizzaro’s reaction
- What happens when methanal undergoes cannizzaro reaction?
- Complete the Following Reactions
- Explain the following reaction: Cannizzaro reaction
- Write the Chemical Equation for the Reaction Involved in Cannizzaro Reaction.
- Complete the Following Reactions
- Write the Product(S) in the Following Reactions -1
- Complete the Following Reaction:
- Complete the Following Reaction:
- Write the Equations Involved in the Following Reactions : Wolff-kishner Reduction
- How do you convert toluene to benzoic acid?
- How Do You Convert the Following : Ethyne to Ethanal
- How do you convert the following: Acetic acid to Methane
- Write the Equations Involved in the Following Reactions : Stephen Reaction
- Write the Structure of 2-hydroxybenzoic Acid.
- Why Carboxylic Acid Does Not Give Reactions of Carbonyl Group?
- Account for the Following: Aromatic Carboxylic Acids Do Not Undergo Friedel-crafts Reaction.
- Name the Reagents Used in the Following Reactions:
- Write the structures of A and B in the following reactions
- An organic compound (A) (molecular formula C8H16O2) was hydrolysed with dilute sulphuric acid to give a carboxylic acid (B) and an alcohol (C). Oxidation of (C) with chromic acid produced (B).
- Name the Reagents Used in the Following Reactions
- Predict the Products of the Following Reactions
- Why Are Alkyl Halides Insoluble in Water?
- Account for the Following: Pka Value of 4-nitrobenzoic Acid is Lower than that of Benzoic Acid.
- Arrange the Following Compounds in Increasing Order of Their Property as Indicated:
- What Happens When Salicylic Acid is Treated with (Ch3co)2 O/H+?
- Why Pka of F-CH2-COOH is Lower than that of Cl−CH2−COOH?
- Write the Reactions Involved Hell-volhard Zelinsky Reaction?
- Account for the Following: Carboxylic Acid is a Stronger Acid than Phenol.
- Although phenoxide ion has more number of resonating structures than carboxylate ion, carboxylic acid is a stronger acid than phenol. Why?
- Distinguish Between the Following : Benzoic Acid and Methyl Benzoate
- Draw the Structures of Major Monohalo Products in Each of the Following Reactions
- Predict the Products of the Following Reactions
- Name the reagents used in the following reactions : CH3-COOH → ClCH2-COOH
- Predict the Products of the Following Reactions
- Give a Reason for the Following : N-n Bond is Weaker than the P-p Bond.
- How will you bring about the following conversion in not more than two steps? Benzoic acid to Benzaldehyde
- Write the Reactions Involved Decarboxylation Reaction
- Convert the following: Propan-1-ol to 2-Bromopropanoic acid
- Assertion (A): Acetic acid but not formic acid, can be halogenated in the presence of red P and Cl2. Reason (R): Acetic acid is a weaker acid than formic acid.
- Account for the following : Cl – CH2COOH is a stronger acid than CH3COOH.
- Write the main product in the following reaction: COONa + NaOH ->[CaO][Delta]
Concepts [25]
- Concepts of Aldehydes, Ketones, and Carboxylic Acids
- Nomenclature of Aldehydes and Ketones
- Nature of Carbonyl Group
- Structure of the Carbonyl Group
- Preparation of Aldehydes and Ketones
- Preparation of Aldehydes
- Preparation of Ketones
- Physical Properties of Aldehydes and Ketones
- Chemical Reactions of Aldehydes and Ketones - Nucleophilic Addition Reactions
- Chemical Reactions of Aldehydes and Ketones - Reduction
- Chemical Reactions of Aldehydes and Ketones - Oxidation
- Chemical Reactions of Aldehydes and Ketones - Reactions Due to α-hydrogen
- Chemical Reactions of Aldehydes and Ketones - Other Reactions
- Uses of Aldehydes and Ketones
- Carboxylic Acids
- Nomenclature of Carboxylic Acids
- Structure of the Carboxyl group
- Methods of Preparation of Carboxylic Acids
- Physical Properties of Carboxylic Acids
- Chemical Reactions of Carboxylic Acids - Reactions Involving Cleavege of O-H Bond
- Chemical Reactions of Carboxylic Acids - Reactions Involving Cleavege of C-OH Bond
- Chemical Reactions of Carboxylic Acids - Reactions Involving –COOH Group
- Chemical Reactions of Carboxylic Acids - Substitution Reactions in the Hydrocarbon Part
- Uses of Carboxylic Acids
- Overview: Aldehydes, Ketones and Carboxylic Acids
