Advertisements
Advertisements
Question
Which of the following does not give Cannizzaro reaction?
Options
(CH3)3C – CHO
(CH3)2 CH – CHO
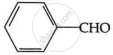
HCHO
Advertisements
Solution
(CH3)2 CH – CHO
Explanation:
Aldehydes which do not have an α-hydrogen atom, undergo self-oxidation and reduction (disproportionation) reaction on heating with concentrated alkali. Such reactions are known as Cannizzaro reactions.
APPEARS IN
RELATED QUESTIONS
Write the product(s) in the following reactions

Complete the following reactions:

How will you convert acetone to acetone cyanohydrin?
complete the following reaction:
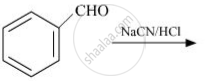
complete the following reaction:
\[\begin{array}{cc}
\phantom{...}\ce{CH3} \\
| \\
\phantom{.................}\ce{CH3-CH-COOH ->[(i) Br2/Red P4][(ii)H2O]}
\end{array}\]
Complete the following reaction:
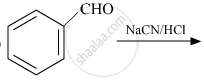
Convert the following:
Benzene to m-nitrobenzaldehyde
Write the chemical reaction involved in Cannizzaro reaction of methanal.
Calcium acetate, on heating, gives:
Benzaldehyde can be prepared by the hydrolysis of:
