Advertisements
Advertisements
Questions
What happens when methanal undergoes cannizzaro reaction?
Give the chemical equation:
What happens when formaldehyde undergoes Cannizzaro’s reaction?
Advertisements
Solution 1
When methanal (formaldehyde, HCHO) undergoes the Cannizzaro reaction, it results in the formation of formic acid HCOOH and methanol CH3OH.
The Cannizzaro reaction is a base-catalyzed disproportionation reaction that occurs with non-alpha-hydrogenated aldehydes (like methanal). In this reaction, one molecule of formaldehyde gets reduced to methanol CH3OH, while the other gets oxidized to formic acid HCOOH.
The reaction can be represented as
\[\ce{2HCHO ->[NaOH] HCOOH + CH3OH}\]
Solution 2
Methanal will undergo Cannizzaro reaction as it possesses α-hydrogen to form methanol.
\[\begin{array}{cc}
\phantom{.}\ce{H}\phantom{..............................}\ce{H}\phantom{.......}\ce{H}\phantom{...........}\phantom{..}\\
\backslash\phantom{..............................}|\phantom{.........}\backslash\phantom{..........}\\
\ce{2C = O + conc{.}KOH -> H - C - OH + C - O- K+}\\
/\phantom{..............................}|\phantom{.........}/\phantom{..........}\\
\ce{\underset{Methanal}{H}}\phantom{.........................}\ce{\underset{Methanol}{H}}\phantom{.}\ce{\underset{methanoate}{\underset{Potassium}{H}}}\phantom{............}\\
\end{array}\]
APPEARS IN
RELATED QUESTIONS
Write the chemical equation for the reaction involved in Cannizzaro reaction.
Write the chemical equations to illustrate the following name reaction:
Cannizzaro’s reaction
Write the product(s) in the following reactions

Write the equation involved in the following reaction:
Etard reaction
Complete the following reactions:

Write the product formed when p-nitro chlorobenzene is heated with aqueous NaOH at 443K followed by acidification?
complete the following reaction:
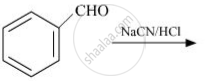
The products obtained in the Cannizzaro reaction are
The key step in cannizzaro reaction in the inter molecular shift qf
Explain the following reaction:
Cannizzaro reaction
