Advertisements
Advertisements
Question
What happens when ethanal is treated with excess ethanol and acid?
Advertisements
Solution
When ethanal reacts with excess ethanol and acid gives acetal.
\[\begin{array}{cc}
\phantom{.......}\ce{CH3}\phantom{.........................}\ce{CH3}\phantom{....}\ce{OC2H5}\\
\phantom{....}\backslash\phantom{..........................}\backslash\phantom{..}/\\
\ce{= O + \underset{(excess)}{C2H5OH} ->[H^⊕]}\\
\phantom{....}/\phantom{..........................}/\phantom{..}\backslash\\
\phantom{.........}\ce{H}\phantom{.........................}\ce{H}\phantom{.....}\ce{OC2H5}\\
\end{array}\]
APPEARS IN
RELATED QUESTIONS
Write the structures of the main products when acetone (CH3 − CO − CH3) reacts with the following reagent :
H2N − NHCONH2/H+
What is meant by the following term? Give an example of the reaction in the following case.
Semicarbazone
Predict the products formed when cyclohexanecarbaldehyde reacts with the following reagents.
Semicarbazide and weak acid
Predict the products formed when cyclohexanecarbaldehyde reacts with the following reagents.
Excess ethanol and acid
How are the following compounds prepared?
acetophenone from benzene
Benzaldehyde can be obtained from benzal chloride. Write reactions for obtaining benzalchloride and then benzaldehyde from it.
Alkenes 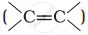 and carbonyl compounds
and carbonyl compounds 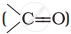 , both contain a π bond but alkenes show electrophilic addition reactions whereas carbonyl compounds show nucleophilic addition reactions. Explain.
, both contain a π bond but alkenes show electrophilic addition reactions whereas carbonyl compounds show nucleophilic addition reactions. Explain.
Draw structures of the given derivatives.
The ethylene ketal of hexan-3-one
Draw structure of the following derivative.
The ethylene ketal of hexan-3-one
Draw structure of the following derivative.
The ethylene ketal of hexan-3-one
