Advertisements
Advertisements
Question
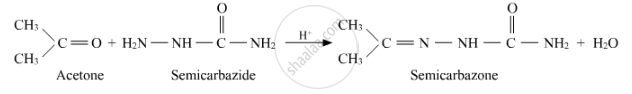
Write the structures of the main products when acetone (CH3 − CO − CH3) reacts with the following reagent :
H2N − NHCONH2/H+
Advertisements
Solution
Acetone reacts with semicarbazide to form semicarbazone.
RELATED QUESTIONS
Draw the structure of the semicarbazone of ethanal.
What is meant by the following term? Give an example of the reaction in the following case.
Hemiacetal
What is meant by the following term? Give an example of the reaction in the following case.
2, 4-DNP-derivative
Predict the products formed when cyclohexanecarbaldehyde reacts with the following reagents.
PhMgBr and then H3O+
Give plausible explanation for the following:
There are two −NH2 groups in semicarbazide. However, only one is involved in the formation of semicarbazones.
Write balanced chemical equations for action of ammonia on - acetaldehyde
Carboxylic acids contain carbonyl group but do not show the nucleophilic addition reaction like aldehydes or ketones. Why?
Why dissociation of HCN is suppressed by the addition of HCL?
Draw structure of the following derivative:
Acetaldehydedimethylacetal
Draw the structure of the following derivative.
Acetaldehydedimethylacetal
