Advertisements
Advertisements
Question
Predict the products formed when cyclohexanecarbaldehyde reacts with the following reagents.
PhMgBr and then H3O+
Advertisements
Solution

APPEARS IN
RELATED QUESTIONS
Write balanced chemical equations for action of ammonia on - formaldehyde
Predict the products of the following reactions:

Predict the product of the following reaction:

Predict the product of the following reaction:
\[\begin{array}{cc}
\phantom{..............}\ce{O}\\
\phantom{..............}||\\
\ce{R - CH = CH - CHO + NH2 - C - NH - NH2 ->[H+]}\end{array}\]
What is meant by the following term? Give an example of the reaction in the following case.
Ketal
Acetaldehyde, Acetone, Di-tert-butyl ketone, Methyl tert-butyl ketone (reactivity towards HCN)
Give plausible explanation for the following:
There are two −NH2 groups in semicarbazide. However, only one is involved in the formation of semicarbazones.
Complete the synthesis by giving missing starting material, reagent or product.
\[\ce{C6H5CHO ->[H2NCONHNH2]}\]
Alkenes 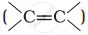 and carbonyl compounds
and carbonyl compounds 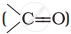 , both contain a π bond but alkenes show electrophilic addition reactions whereas carbonyl compounds show nucleophilic addition reactions. Explain.
, both contain a π bond but alkenes show electrophilic addition reactions whereas carbonyl compounds show nucleophilic addition reactions. Explain.
The pH of blood does not appreciably change by a small addition of acid or base because
The most stable reagent for the conversion of R – CH2OH → RCHO is
What happens when propanone is treated with CH3MgBr and then hydrolysed?
What happens when ethanal is treated with excess ethanol and acid?
Draw structure of the following derivative.
Acetaldehydedimethylacetal
Draw structure of the following derivative.
The ethylene ketal of hexan-3-one
Draw structures of the following derivatives.
Acetaldehydedimethylacetal
Draw the structure of the following derivative.
Acetaldehydedimethylacetal
Draw the structure of the given derivative.
The ethylene ketal of hexan-3-one
