Advertisements
Advertisements
Question
How will you bring about the following conversion in not more than two steps?
Bromobenzene to 1-Phenylethanol
Advertisements
Solution
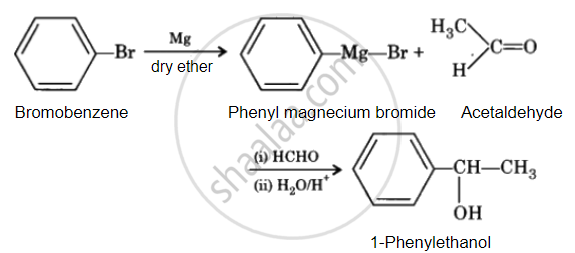
APPEARS IN
RELATED QUESTIONS
Write balanced chemical equations for action of ammonia on - formaldehyde
Draw the structure of the semicarbazone of ethanal.
Predict the product of the following reaction:
\[\begin{array}{cc}
\phantom{..............}\ce{O}\\
\phantom{..............}||\\
\ce{R - CH = CH - CHO + NH2 - C - NH - NH2 ->[H+]}\end{array}\]
What is meant by the following term? Give an example of the reaction in the following case.
Semicarbazone
Predict the products formed when cyclohexanecarbaldehyde reacts with the following reagents.
PhMgBr and then H3O+
Explain the mechanism of alkaline hydrolysis of tert-butyl bromide with energy profile diagram.
How will you convert sodium acetate to methane?
How will you convert benzoic acid to m-bromobenzoic acid?
Identify the compounds A, B and C in the following reaction.
\[\ce{CH3 - Br ->[Mg/ether] (A) ->[(i) CO][(ii) Water] (B) ->[CH3OH/H+][Δ] (C)}\]
Which one of the following gives only one monochloro derivative?
Which of the following has the most acidic hydrogen?
Paraldehyde is formed as a result of polymerisation:-
The most stable reagent for the conversion of R – CH2OH → RCHO is
Draw the structure of the following derivative.
The ethylene ketal of hexane-3-one
Draw structures of the following derivative.
The ethylene ketal of hexan-3-one
Draw the structure of the following derivative.
The ethylene ketal of hexan-3-one
Draw structures of the given derivatives.
The ethylene ketal of hexan-3-one
Draw structure of the following derivative.
The ethylene ketal of hexan-3-one
Give an example of the reaction in the following case.
Oxime
