Advertisements
Advertisements
प्रश्न
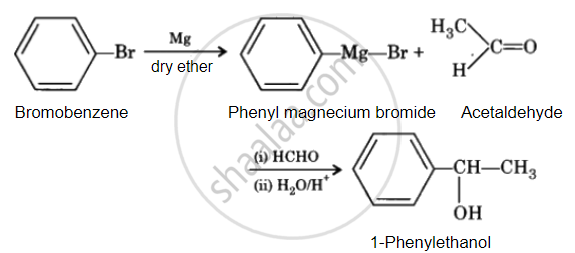
How will you bring about the following conversion in not more than two steps?
Bromobenzene to 1-Phenylethanol
Advertisements
उत्तर
APPEARS IN
संबंधित प्रश्न
Predict the product of the following reaction:

What is meant by the following term? Give an example of the reaction in the following case.
Cyanohydrin
What is meant by the following term? Give an example of the reaction in the following case.
Semicarbazone
Complete the synthesis by giving missing starting material, reagent or product.
\[\ce{C6H5CHO ->[H2NCONHNH2]}\]
How are the following compounds prepared?
acetophenone from benzene
Write balanced chemical equations for action of ammonia on - acetaldehyde
Write balanced chemical equations for action of ammonia on - acetone
Give a simple chemical test to distinguish between 
Write the main product formed when propanal reacts with the following reagents:
H2N- NH2 followed by heating with KOH in ethylene glycol.
Write the name of product formed, when acetone is treated with 2, 4-dinitrophenyl hydrazine.
What is the action of sodium hypoiodite on acetone?
What happens when propanone is treated with CH3MgBr and then hydrolysed?
What happens when ethanal is treated with excess ethanol and acid?
The increasing order of the following compounds towards HCN addition is:
| (i) |  |
| (ii) |  |
| (iii) |  |
| (iv) |  |
Draw structure of the following derivative.
The ethylene ketal of hexane-3-one
Draw structure of the following derivative.
Acetaldehydedimethylacetal
Draw structure of the following derivative:
Acetaldehydedimethylacetal
Give an example of the reaction in the following case.
Imine
