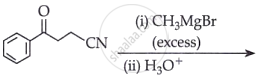Advertisements
Advertisements
प्रश्न
Write balanced chemical equations for action of ammonia on - formaldehyde
Advertisements
उत्तर
a. 6HCHO + 4NH3 → (CH2)6N4 + 6H2O
Formaldehyde Urotropine
APPEARS IN
संबंधित प्रश्न
Predict the products of the following reactions:

Draw the structure of the semicarbazone of ethanal.
Predict the product of the following reaction:

Predict the product of the following reaction:

What is meant by the following term? Give an example of the reaction in the following case.
Acetal
What is meant by the following term? Give an example of the reaction in the following case.
Hemiacetal
What is meant by the following term? Give an example of the reaction in the following case.
Schiff’s base
Predict the products formed when cyclohexanecarbaldehyde reacts with the following reagents.
PhMgBr and then H3O+
Predict the products formed when cyclohexanecarbaldehyde reacts with the following reagents.
Excess ethanol and acid
Write balanced chemical equations for action of ammonia on - acetone
What is the action of the following reagents on ethanoic acid?
1) `LiAlH_4"/"H_3O^+`
2) `PCl_3 , "heat"`
3) `P_2O_5, "heat"`
Write the structure of Phenylmethanamine.
How will you convert sodium acetate to methane?
Give a simple chemical test to distinguish between 
Write the main product formed when propanal reacts with the following reagents:
2 moles of 3 CH OH in presence of dry HCl
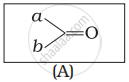
Which of the following is the correct representation for intermediate of nucleophilic addition reaction to the given carbonyl compound (A):
(i) 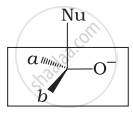
(ii) 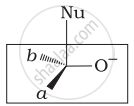
(iii) 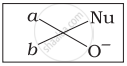
(iv) 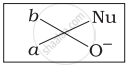
Benzaldehyde can be obtained from benzal chloride. Write reactions for obtaining benzalchloride and then benzaldehyde from it.
Identify the compounds A, B and C in the following reaction.
\[\ce{CH3 - Br ->[Mg/ether] (A) ->[(i) CO][(ii) Water] (B) ->[CH3OH/H+][Δ] (C)}\]
Reaction of aqueous sodium hydroxide on chlorobenzene gives which of the following products?
A Idol condensation will not be observed in
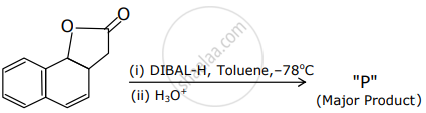
The product "P" in the above reaction is:
The increasing order of the following compounds towards HCN addition is:
| (i) |  |
| (ii) |  |
| (iii) |  |
| (iv) |  |
In the following reaction
\[\ce{Carbonyl compound + MeOH <=>[HCl] acetal}\]
Rate of the reaction is the highest for ______.
Draw structure of the following derivative:
Acetaldehydedimethylacetal
Draw structure of the following derivative.
The ethylene ketal of hexan-3-one
The major product of the following reaction is: