Advertisements
Advertisements
प्रश्न
How will you convert sodium acetate to methane?
Convert sodium acetate to methane.
Advertisements
उत्तर
When sodium acetate is heated with soda lime (NaOH + CaO), methane is obtained.
\[\begin{array}{cc}
\ce{O}\phantom{.................................}\\
||\phantom{.................................}\\
\ce{\underset{(Sodium acetate)}{CH3 - C - ONa} + NaOH ->[CaO][\Delta] \underset{(Methane)}{CH4} + Na2CO3}\\
\end{array}\]
APPEARS IN
संबंधित प्रश्न
Predict the products of the following reactions:

Draw the structure of the semicarbazone of ethanal.
Arrange the following compound in increasing order of its reactivity in nucleophilic addition reactions.
Ethanal, Propanal, Propanone, Butanone.
Hint: Consider steric effect and electronic effect.
Predict the product of the following reaction:
\[\begin{array}{cc}
\phantom{..............}\ce{O}\\
\phantom{..............}||\\
\ce{R - CH = CH - CHO + NH2 - C - NH - NH2 ->[H+]}\end{array}\]
What is meant by the following term? Give an example of the reaction in the following case.
Schiff’s base
Predict the products formed when cyclohexanecarbaldehyde reacts with the following reagents.
PhMgBr and then H3O+
Predict the products formed when cyclohexanecarbaldehyde reacts with the following reagents.
Semicarbazide and weak acid
Give plausible explanation for the following:
There are two −NH2 groups in semicarbazide. However, only one is involved in the formation of semicarbazones.
Explain the mechanism of alkaline hydrolysis of tert-butyl bromide with energy profile diagram.
How will you convert benzoic acid to m-bromobenzoic acid?
Alkenes 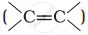 and carbonyl compounds
and carbonyl compounds 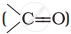 , both contain a π bond but alkenes show electrophilic addition reactions whereas carbonyl compounds show nucleophilic addition reactions. Explain.
, both contain a π bond but alkenes show electrophilic addition reactions whereas carbonyl compounds show nucleophilic addition reactions. Explain.
Reaction of aqueous sodium hydroxide on chlorobenzene gives which of the following products?
Which one of the following gives only one monochloro derivative?
What is the action of sodium hypoiodite on acetone?
Arrange the following in the increasing order of their property indicated:
Ethanal, Propanone, Propanal, Butanone (reactivity towards nucleophilic addition)
What happens when ethanal is treated with excess ethanol and acid?
Aldehydes and ketones react with hydroxylamine to form ______.
Draw structures of the given derivatives.
The ethylene ketal of hexan-3-one
Draw structure of the following derivative.
The ethylene ketal of hexan-3-one
The product of the following reaction is
\[\begin{array}{cc}
\ce{O}\phantom{.........}\\
||\phantom{.........}\\
\ce{C2H5 - C - CH3 ->[H2/Ni][\Delta] \phantom{..}?}\end{array}\]
Draw the structure of the following derivative.
The ethylene ketal of hexan-3-one
Draw structures of the following derivatives.
Acetaldehydedimethylacetal
Draw structure of the following derivative.
The ethylene ketal of hexan-3-one
Draw the structure of the following derivative.
Acetaldehydedimethylacetal
Draw structures of the given derivatives.
The ethylene ketal of hexan-3-one
