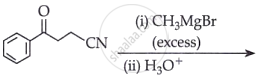Advertisements
Advertisements
प्रश्न
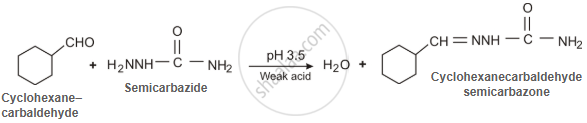
Predict the products formed when cyclohexanecarbaldehyde reacts with the following reagents.
Semicarbazide and weak acid
Advertisements
उत्तर
APPEARS IN
संबंधित प्रश्न
Write the structures of the main products when acetone (CH3 − CO − CH3) reacts with the following reagent :
H2N − NHCONH2/H+
What is meant by the following term? Give an example of the reaction in the following case.
Cyanohydrin
What is meant by the following term? Give an example of the reaction in the following case.
Acetal
How will you convert sodium acetate to methane?
Give a simple chemical test to distinguish between 
Identify the compounds A, B and C in the following reaction.
\[\ce{CH3 - Br ->[Mg/ether] (A) ->[(i) CO][(ii) Water] (B) ->[CH3OH/H+][Δ] (C)}\]
A Idol condensation will not be observed in
What happens when ethanal is treated with excess ethanol and acid?
Which will undergo faster nucleophilic addition reaction?
Acetaldehyde or Propanone
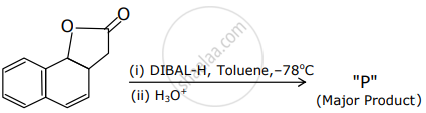
The product "P" in the above reaction is:
The product of following reaction is
\[\ce{CH3 - CH = CH - CH2 - CHO ->[i) LiAlH4][ii) H3O+]}\] ______?
Out of p-tolualdehyde and p-nitrobenzaldehyde, which one is more reactive towards nucleophilic addition reactions, why?
Write the structure of the product formed when acetone reacts with 2, 4 DNP reagent.
Draw structure of the following derivative.
The ethylene ketal of hexane-3-one
The product of the following reaction is
\[\begin{array}{cc}
\ce{O}\phantom{.........}\\
||\phantom{.........}\\
\ce{C2H5 - C - CH3 ->[H2/Ni][\Delta] \phantom{..}?}\end{array}\]
Draw the structure of the following derivative.
The ethylene ketal of hexan-3-one
Draw structure of the following derivative:
Acetaldehydedimethylacetal
Draw structure of the following derivative.
The ethylene ketal of hexan-3-one
Draw structure of the following derivative.
The ethylene ketal of hexan-3-one
The major product of the following reaction is: