Advertisements
Advertisements
प्रश्न
Write the main product formed when propanal reacts with the following reagents:
H2N- NH2 followed by heating with KOH in ethylene glycol.
Advertisements
उत्तर
\[\ce{CH3CH2CHO + H2N - NH2 -> CH3CH2CH = N - NH2 + H2O}\]
This is a nucleophilic addition-elimination process where the carbonyl group (C = O) is replaced with a hydrazone group \[\ce{(C = N - NH2)}\].
The hydrazone is then heated with KOH in ethylene glycol, a strong base under high-temperature conditions. This leads to the loss of nitrogen gas (N2) and the reduction of the carbon–nitrogen double bond to a C–H bond.
\[\ce{CH3CH2CH = N - NH2 \overset{KOH/ethylene glycol,Δ}{->} CH3CH2CH3 + N2 ↑}\]
APPEARS IN
संबंधित प्रश्न
Arrange the following compound in increasing order of its reactivity in nucleophilic addition reactions.
Benzaldehyde, p-Tolualdehyde, p-Nitrobenzaldehyde, Acetophenone.
Hint: Consider steric effect and electronic effect.
What is meant by the following term? Give an example of the reaction in the following case.
2, 4-DNP-derivative
Give plausible explanation for the following:
There are two −NH2 groups in semicarbazide. However, only one is involved in the formation of semicarbazones.
Complete the synthesis by giving missing starting material, reagent or product.

Identify the compounds A, B and C in the following reaction.
\[\ce{CH3 - Br ->[Mg/ether] (A) ->[(i) CO][(ii) Water] (B) ->[CH3OH/H+][Δ] (C)}\]
A Idol condensation will not be observed in
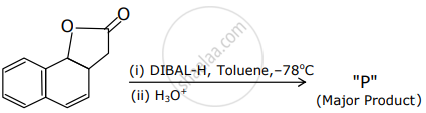
The product "P" in the above reaction is:
Draw the structure of the following derivative.
The ethylene ketal of hexan-3-one
Draw structure of the following derivative.
The ethylene ketal of hexan-3-one
Complete the following reaction:
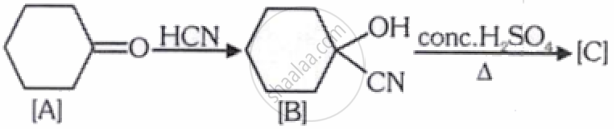
[C] is __________.
