Advertisements
Advertisements
प्रश्न
What is meant by the following term? Give an example of the reaction in the following case.
Imine
Advertisements
उत्तर
Imines are formed when carbonyl compounds react with ammonia derivatives.
\[\begin{array}{cc}
\backslash\phantom{......................}\backslash\phantom{................}\\
\ce{C = O + H2N - Z -> \phantom{.}= N-Z + H2O}\\
\ce{\underset{Compound}{\underset{Carbonyl}{/}}}\phantom{.................}\ce{\underset{Imine}{/}}\phantom{..................}
\end{array}\]
Z = R, 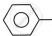 , −NH2, −OH, etc.
, −NH2, −OH, etc.
APPEARS IN
संबंधित प्रश्न
Predict the products of the following reactions:

Write the products formed when CH3CHO reacts with the following reagents : HCN
Write the products formed when CH3CHO reacts with the following reagents : H2N – OH
Predict the products formed when cyclohexanecarbaldehyde reacts with the following reagents.
PhMgBr and then H3O+
Acetaldehyde, Acetone, Di-tert-butyl ketone, Methyl tert-butyl ketone (reactivity towards HCN)
How will you bring about the following conversion in not more than two steps?
Bromobenzene to 1-Phenylethanol
Complete the synthesis by giving missing starting material, reagent or product.
\[\ce{C6H5CHO ->[H2NCONHNH2]}\]
Complete the synthesis by giving missing starting material, reagent or product.

How are the following compounds prepared?
benzaldehyde from benzoyl chloride
Acetone, Acetaldehyde, Benzaldehyde, Acetophenone – reactivity towards addition of HCN.
The pH of blood does not appreciably change by a small addition of acid or base because
Which one of the following gives only one monochloro derivative?
Grignard reagent on reaction with acetone forms.
The most stable reagent for the conversion of R – CH2OH → RCHO is
Arrange the following in the increasing order of their property indicated:
Ethanal, Propanone, Propanal, Butanone (reactivity towards nucleophilic addition)
Why dissociation of HCN is suppressed by the addition of HCL?
Draw structures of the following derivative.
The ethylene ketal of hexan-3-one
Draw the structure of the following derivative.
Acetaldehydedimethylacetal
Give an example of the reaction in the following case.
Oxime
