Advertisements
Advertisements
प्रश्न
Write a test to differentiate between pentan-2-one and pentan-3-one.
Advertisements
उत्तर
Pentan--one having a group forms a yellow ppt. of iodoform with an alkaline solution of iodine (i.e., iodoform test) while pentan-3-one does not.
\[\begin{array}{cc}
\ce{O}\phantom{.....................................}\\
||\phantom{.....................................}\\
\ce{CH3CH2\underset{Pentan-2-one}{CH2 - C - CH3} ->[I2/NaOH][(Iodoform test)] \underset{Yellow ppt.}{CHI3} + \underset{Sodium butanoate}{CH3CH2CH2COONa}}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
Write balanced chemical equations for action of ammonia on - formaldehyde
Predict the product of the following reaction:
\[\begin{array}{cc}
\phantom{..............}\ce{O}\\
\phantom{..............}||\\
\ce{R - CH = CH - CHO + NH2 - C - NH - NH2 ->[H+]}\end{array}\]
What is meant by the following term? Give an example of the reaction in the following case.
Ketal
Acetaldehyde, Acetone, Di-tert-butyl ketone, Methyl tert-butyl ketone (reactivity towards HCN)
Complete the synthesis by giving missing starting material, reagent or product.
\[\ce{C6H5CHO ->[H2NCONHNH2]}\]
Write the structure of Phenylmethanamine.
What are amines?
Grignard reagent on reaction with acetone forms.
Acetaldehyde and acetone differ in their reaction with
Write the name of product formed, when acetone is treated with 2, 4-dinitrophenyl hydrazine.
Arrange the following in the increasing order of their property indicated:
Ethanal, Propanone, Propanal, Butanone (reactivity towards nucleophilic addition)
What happens when ethanal is treated with excess ethanol and acid?
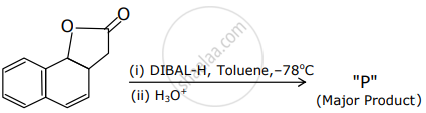
The product "P" in the above reaction is:
In the following reaction
\[\ce{Carbonyl compound + MeOH <=>[HCl] acetal}\]
Rate of the reaction is the highest for ______.
Which of the following is most reactive in nucleophilic addition reactions?
Draw structure of the following derivative.
Acetaldehydedimethylacetal
Draw the structure of the following derivative.
The ethylene ketal of hexan-3-one
Draw the structure of the given derivative.
The ethylene ketal of hexan-3-one
