Advertisements
Advertisements
Question
Which will undergo faster nucleophilic addition reaction?
Acetaldehyde or Propanone
Advertisements
Solution
Aldehydes are generally more reactive than ketones in nucleophilic addition reactions due to steric hindrance. As the electron density at the carbonyl carbon increases, the +I effect increases, which decreases the chances of attack by a nucleophile. Thus, acetaldehyde is more reactive than propanone.
APPEARS IN
RELATED QUESTIONS
Acetaldehyde, when treated with which among the following reagents does NOT undergo addition reaction?
(A) Ammonia
(B) Hydroxylamine
(C) Ammoniacal silver nitrate
(D) Semicarbazide
Write the products formed when CH3CHO reacts with the following reagents : HCN
Predict the products formed when cyclohexanecarbaldehyde reacts with the following reagents.
PhMgBr and then H3O+
Predict the products formed when cyclohexanecarbaldehyde reacts with the following reagents.
Semicarbazide and weak acid
Complete the synthesis by giving missing starting material, reagent or product.

Which of the following is the correct representation for intermediate of nucleophilic addition reaction to the given carbonyl compound (A):
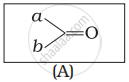
(i) 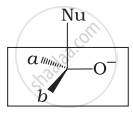
(ii) 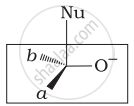
(iii) 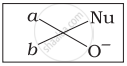
(iv) 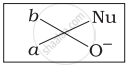
Arrange the following in the increasing order of their property indicated:
Ethanal, Propanone, Propanal, Butanone (reactivity towards nucleophilic addition)
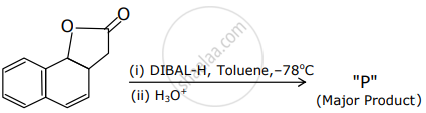
The product "P" in the above reaction is:
Draw structure of the following derivative.
The ethylene ketal of hexan-3-one
Draw the structure of the following derivative.
Acetaldehydedimethylacetal
