Advertisements
Advertisements
Question
Give plausible explanation for the following:
There are two −NH2 groups in semicarbazide. However, only one is involved in the formation of semicarbazones.
Advertisements
Solution
Semicarbazide only has one of the two −NH2 groups resonance and this group is directly attached to the carbonyl-carbon atom.
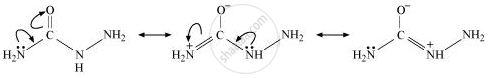
Therefore, the electron density on −NH2 group involved in the resonance also decreases. As a result, it cannot act as a nucleophile. Since the other −NH2 group is not involved in resonance, it can act as a nucleophile and attack carbonyl-carbon atoms of aldehydes and ketones to produce semicarbazones.
APPEARS IN
RELATED QUESTIONS
How are the following compounds prepared?
acetophenone from benzene
Write balanced chemical equations for action of ammonia on - acetaldehyde
Write balanced chemical equations for action of ammonia on - acetone
Write the structure of Phenylmethanamine.
Write the main product formed when propanal reacts with the following reagents:
H2N- NH2 followed by heating with KOH in ethylene glycol.
Acetone, Acetaldehyde, Benzaldehyde, Acetophenone – reactivity towards addition of HCN.
How will you convert benzoic acid to m-bromobenzoic acid?
Carboxylic acids contain carbonyl group but do not show the nucleophilic addition reaction like aldehydes or ketones. Why?
The pH of blood does not appreciably change by a small addition of acid or base because
A Idol condensation will not be observed in
Acetaldehyde and acetone differ in their reaction with
Paraldehyde is formed as a result of polymerisation:-
Write the name of product formed, when acetone is treated with 2, 4-dinitrophenyl hydrazine.
Arrange the following in the increasing order of their property indicated:
Ethanal, Propanone, Propanal, Butanone (reactivity towards nucleophilic addition)
Aldehydes and ketones react with hydroxylamine to form ______.
The product of the following reaction is
\[\begin{array}{cc}
\ce{O}\phantom{.........}\\
||\phantom{.........}\\
\ce{C2H5 - C - CH3 ->[H2/Ni][\Delta] \phantom{..}?}\end{array}\]
Draw structure of the following derivative:
Acetaldehydedimethylacetal
Draw structure of the following derivative.
The ethylene ketal of hexan-3-one
Which of the following will not be formed when calcium formate is distilled with calcium acetate?
Complete the following reaction:
[C] is __________.
