Advertisements
Advertisements
प्रश्न
Give plausible explanation for the following:
There are two −NH2 groups in semicarbazide. However, only one is involved in the formation of semicarbazones.
Advertisements
उत्तर
Semicarbazide only has one of the two −NH2 groups resonance and this group is directly attached to the carbonyl-carbon atom.
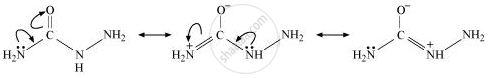
Therefore, the electron density on −NH2 group involved in the resonance also decreases. As a result, it cannot act as a nucleophile. Since the other −NH2 group is not involved in resonance, it can act as a nucleophile and attack carbonyl-carbon atoms of aldehydes and ketones to produce semicarbazones.
APPEARS IN
संबंधित प्रश्न
Predict the product of the following reaction:
\[\begin{array}{cc}
\phantom{..............}\ce{O}\\
\phantom{..............}||\\
\ce{R - CH = CH - CHO + NH2 - C - NH - NH2 ->[H+]}\end{array}\]
What is meant by the following term? Give an example of the reaction in the following case.
Cyanohydrin
What is meant by the following term? Give an example of the reaction in the following case.
Imine
Predict the products formed when cyclohexanecarbaldehyde reacts with the following reagents.
Excess ethanol and acid
Give plausible explanation for the following:
Cyclohexanone forms cyanohydrin in good yield but 2, 2, 6 trimethylcyclohexanone does not.
What are amines?
How will you convert sodium acetate to methane?
Reaction of aqueous sodium hydroxide on chlorobenzene gives which of the following products?
Which one of the following gives only one monochloro derivative?
Which of the following has the most acidic hydrogen?
Paraldehyde is formed as a result of polymerisation:-
The most stable reagent for the conversion of R – CH2OH → RCHO is
Arrange the following in the increasing order of their property indicated:
Ethanal, Propanone, Propanal, Butanone (reactivity towards nucleophilic addition)
In the following reaction
\[\ce{Carbonyl compound + MeOH <=>[HCl] acetal}\]
Rate of the reaction is the highest for ______.
Aldehydes and ketones react with hydroxylamine to form ______.
Out of p-tolualdehyde and p-nitrobenzaldehyde, which one is more reactive towards nucleophilic addition reactions, why?
Draw structures of the following derivative.
The ethylene ketal of hexan-3-one
Draw structure of the following derivative.
The ethylene ketal of hexan-3-one
Draw the structure of the following derivative.
Acetaldehydedimethylacetal
Draw the structure of the given derivative.
The ethylene ketal of hexan-3-one
