Advertisements
Advertisements
प्रश्न
Give plausible explanation for the following:
During the preparation of esters from a carboxylic acid and an alcohol in the presence of an acid catalyst, the water or the ester should be removed as soon as it is formed.
Advertisements
उत्तर
Ester, along with water, is formed reversibly from a carboxylic acid and an alcohol in the presence of an acid.
\[\ce{\underset{Carboxylic acid}{RCOOH} + \underset{Alcohol}{R'OH} <=>[H2SO4] \underset{Ester}{RCOOR'} + H2O}\]
If either water or ester is not removed as soon as it is formed, it reacts to give back the reactants, as the reaction is reversible. Therefore, either of the two should be removed to shift the equilibrium in the forward direction, i.e., to produce more ester.
APPEARS IN
संबंधित प्रश्न
Explain the mechanism of esterification
Name the reagents used in the following reactions:

Draw the structures of major monohalo products in each of the following reactions :

Complete the synthesis by giving missing starting material, reagent or product.
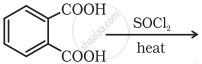
Which of the following carboxylic acid can be esterified most readily?
Carboxylic acid reacts with diazomethane to yield:
Acid anhydride on reaction with primary amine gives compound having a functional group ______.
Write the equations for the following reaction:
Salicylic acid is treated with acetic anhydride in the presence of conc H2SO4
Given below are two statements:
Statement I: The esterification of carboxylic acid with an alcohol is a nucleophilic acyl substitution.
Statement II: Electron withdrawing groups in the carboxylic acid will increase the rate of esterification reactions.
Choose the most appropriate options.
In the given reaction,

The number of n electrons present in the product 'P' is ______.
A reaction of ethyl amine and acetic anhydride leads to the formation of ______.
\[\begin{array}{cc}
\phantom{}\ce{\underset{}{CH3CH2COOH} ->[Cl2][ Red P] \underset{}{A}->[Alc KOH] \underset{}{B}}\\
\end{array}\]
What is (B):
