Advertisements
Advertisements
Question
Draw the structure of the semicarbazone of ethanal.
Advertisements
Solution
\[\begin{array}{cc}
\ce{H3C}\phantom{.........................}\ce{O}\phantom{..................}\ce{H}\phantom{..............}\ce{O}\phantom{.............}\\
\phantom{..}\backslash\phantom{........................}||\phantom{...................}|\phantom{...............}||\phantom{...........}\\
\ce{\underset{Ethanal}{C = O} + H2N - NH - C - NH2 -> \underset{Semicarbazone}{CH3 - C = N - NH - C - NH2}}\\
/\phantom{.....................................................................}\\
\ce{H}\phantom{........................................................................}\\
\end{array}\]
APPEARS IN
RELATED QUESTIONS
Write balanced chemical equations for action of ammonia on - formaldehyde
How are the following compounds prepared?
benzaldehyde from benzene
Arrange the following compound in increasing order of its reactivity in nucleophilic addition reactions.
Benzaldehyde, p-Tolualdehyde, p-Nitrobenzaldehyde, Acetophenone.
Hint: Consider steric effect and electronic effect.
Give plausible explanation for the following:
There are two −NH2 groups in semicarbazide. However, only one is involved in the formation of semicarbazones.
Complete the synthesis by giving missing starting material, reagent or product.
\[\ce{C6H5CHO ->[H2NCONHNH2]}\]
How are the following compounds prepared?
acetophenone from benzene
Alkenes 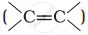 and carbonyl compounds
and carbonyl compounds 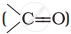 , both contain a π bond but alkenes show electrophilic addition reactions whereas carbonyl compounds show nucleophilic addition reactions. Explain.
, both contain a π bond but alkenes show electrophilic addition reactions whereas carbonyl compounds show nucleophilic addition reactions. Explain.
In the following reaction
\[\ce{Carbonyl compound + MeOH <=>[HCl] acetal}\]
Rate of the reaction is the highest for ______.
Draw structure of the following derivative:
Acetaldehydedimethylacetal
Which of the following will not be formed when calcium formate is distilled with calcium acetate?
