Advertisements
Advertisements
Question
How are the following compounds prepared?
benzaldehyde from benzene
Advertisements
Solution
(a) Benzaldehyde from benzene

Benzene
Gatterman Koch synthesis
APPEARS IN
RELATED QUESTIONS
Write balanced chemical equations for action of ammonia on - formaldehyde
Write the structures of the main products when acetone (CH3 − CO − CH3) reacts with the following reagent :
H2N − NHCONH2/H+
Write the products formed when CH3CHO reacts with the following reagents : HCN
Arrange the following compound in increasing order of its reactivity in nucleophilic addition reactions.
Ethanal, Propanal, Propanone, Butanone.
Hint: Consider steric effect and electronic effect.
Predict the product of the following reaction:

What is meant by the following term? Give an example of the reaction in the following case.
Cyanohydrin
What is meant by the following term? Give an example of the reaction in the following case.
Hemiacetal
Complete the synthesis by giving missing starting material, reagent or product.
\[\ce{C6H5CHO ->[H2NCONHNH2]}\]
Explain the mechanism of alkaline hydrolysis of tert-butyl bromide with energy profile diagram.
How are the following compounds prepared?
acetophenone from benzene
Write balanced chemical equations for action of ammonia on - acetaldehyde
Write balanced chemical equations for action of ammonia on - acetone
Write the structure of Phenylmethanamine.
Give a simple chemical test to distinguish between 
Write the main product formed when propanal reacts with the following reagents:
2 moles of 3 CH OH in presence of dry HCl
How will you convert benzoic acid to m-bromobenzoic acid?
The pH of blood does not appreciably change by a small addition of acid or base because
Which one of the following gives only one monochloro derivative?
Grignard reagent on reaction with acetone forms.
A Idol condensation will not be observed in
Acetaldehyde and acetone differ in their reaction with
Paraldehyde is formed as a result of polymerisation:-
Which among the following is most reactive to give nucleophilic addition?
Write the name of product formed, when acetone is treated with 2, 4-dinitrophenyl hydrazine.
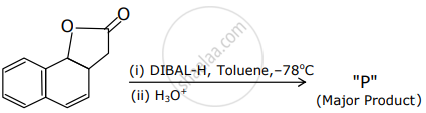
The product "P" in the above reaction is:
The product of following reaction is
\[\ce{CH3 - CH = CH - CH2 - CHO ->[i) LiAlH4][ii) H3O+]}\] ______?
Draw structure of the following derivative.
Acetaldehydedimethylacetal
Complete the following reaction:
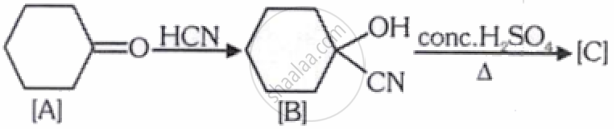
[C] is __________.
