Advertisements
Advertisements
Question
Complete the synthesis by giving missing starting material, reagent or product.
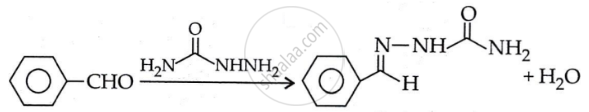
\[\ce{C6H5CHO ->[H2NCONHNH2]}\]
Advertisements
Solution 1
\[\begin{array}{cc}
\phantom{....................................}\ce{O}\\
\phantom{....................................}||\\
\ce{\underset{Benzaldehyde}{C6H5CHO} + \underset{Semicarbazide}{H2NCONHNH2} -> C6H5CH \underset{Benzaldehyde semicarbazide}{= NNHC - NH2} + H2O}\end{array}\]
Solution 2
APPEARS IN
RELATED QUESTIONS
Acetaldehyde, when treated with which among the following reagents does NOT undergo addition reaction?
(A) Ammonia
(B) Hydroxylamine
(C) Ammoniacal silver nitrate
(D) Semicarbazide
Arrange the following in the increasing order of their reactivity towards nucleophilic addition reaction:
C6H5COCH3, CH3-CHO, CH3COCH3
Write the products formed when CH3CHO reacts with the following reagents : H2N – OH
Predict the products formed when cyclohexanecarbaldehyde reacts with the following reagents.
PhMgBr and then H3O+
Explain the mechanism of alkaline hydrolysis of tert-butyl bromide with energy profile diagram.
What is the action of the following reagents on ethanoic acid?
1) `LiAlH_4"/"H_3O^+`
2) `PCl_3 , "heat"`
3) `P_2O_5, "heat"`
How will you convert sodium acetate to methane?
Reaction of aqueous sodium hydroxide on chlorobenzene gives which of the following products?
Which one of the following gives only one monochloro derivative?
A Idol condensation will not be observed in
Paraldehyde is formed as a result of polymerisation:-
Which among the following is most reactive to give nucleophilic addition?
Write the name of product formed, when acetone is treated with 2, 4-dinitrophenyl hydrazine.
What happens when propanone is treated with CH3MgBr and then hydrolysed?
In the following reaction
\[\ce{Carbonyl compound + MeOH <=>[HCl] acetal}\]
Rate of the reaction is the highest for ______.
Which of the following is most reactive in nucleophilic addition reactions?
Out of p-tolualdehyde and p-nitrobenzaldehyde, which one is more reactive towards nucleophilic addition reactions, why?
Draw structures of the given derivatives.
The ethylene ketal of hexan-3-one
Why dissociation of HCN is suppressed by the addition of HCL?
Draw the structure of the following derivative.
Acetaldehydedimethylacetal
