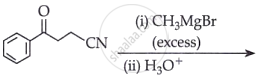Advertisements
Advertisements
Question
Arrange the following compound in increasing order of its reactivity in nucleophilic addition reactions.
Benzaldehyde, p-Tolualdehyde, p-Nitrobenzaldehyde, Acetophenone.
Hint: Consider steric effect and electronic effect.
Advertisements
Solution
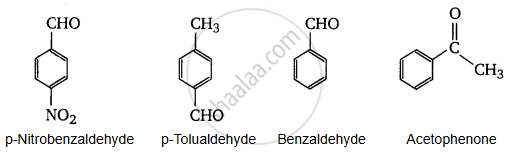
- The +I effect is more in ketones than in aldehydes. Hence, acetophenone is the least reactive in nucleophilic addition reactions.
- Among aldehydes, the +I effect is the highest in p-tolualdehyde because of the presence of the electron-donating −CH3 group and the lowest in p-nitrobezaldehyde because of the presence of the electron-withdrawing −NO2 group.
- Hence, the increasing order of the reactivities of the given compounds is:
Acetophenone < p-Tolualdehyde < Benzaldehyde < p-Nitrobenzaldehyde
APPEARS IN
RELATED QUESTIONS
Predict the product of the following reaction:

Predict the product of the following reaction:
\[\begin{array}{cc}
\phantom{..............}\ce{O}\\
\phantom{..............}||\\
\ce{R - CH = CH - CHO + NH2 - C - NH - NH2 ->[H+]}\end{array}\]
What is meant by the following term? Give an example of the reaction in the following case.
Semicarbazone
Give plausible explanation for the following:
There are two −NH2 groups in semicarbazide. However, only one is involved in the formation of semicarbazones.
How are the following compounds prepared?
benzaldehyde from benzoyl chloride
What is the action of the following reagents on ethanoic acid?
1) `LiAlH_4"/"H_3O^+`
2) `PCl_3 , "heat"`
3) `P_2O_5, "heat"`
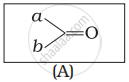
Which of the following is the correct representation for intermediate of nucleophilic addition reaction to the given carbonyl compound (A):
(i) 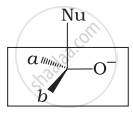
(ii) 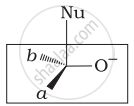
(iii) 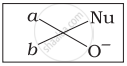
(iv) 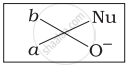
Write a test to differentiate between pentan-2-one and pentan-3-one.
Benzaldehyde can be obtained from benzal chloride. Write reactions for obtaining benzalchloride and then benzaldehyde from it.
The pH of blood does not appreciably change by a small addition of acid or base because
Acetaldehyde and acetone differ in their reaction with
Draw structures of the given derivatives.
The ethylene ketal of hexan-3-one
Draw structure of the following derivative.
Acetaldehydedimethylacetal
Why dissociation of HCN is suppressed by the addition of HCL?
Draw the structure of the following derivative.
Acetaldehydedimethylacetal
Draw structures of the following derivatives.
Acetaldehydedimethylacetal
Draw structure of the following derivative.
The ethylene ketal of hexan-3-one
Draw the structure of the given derivative.
The ethylene ketal of hexan-3-one
The major product of the following reaction is: