Advertisements
Advertisements
Question
What is meant by the following term? Give an example of the reaction in the following case.
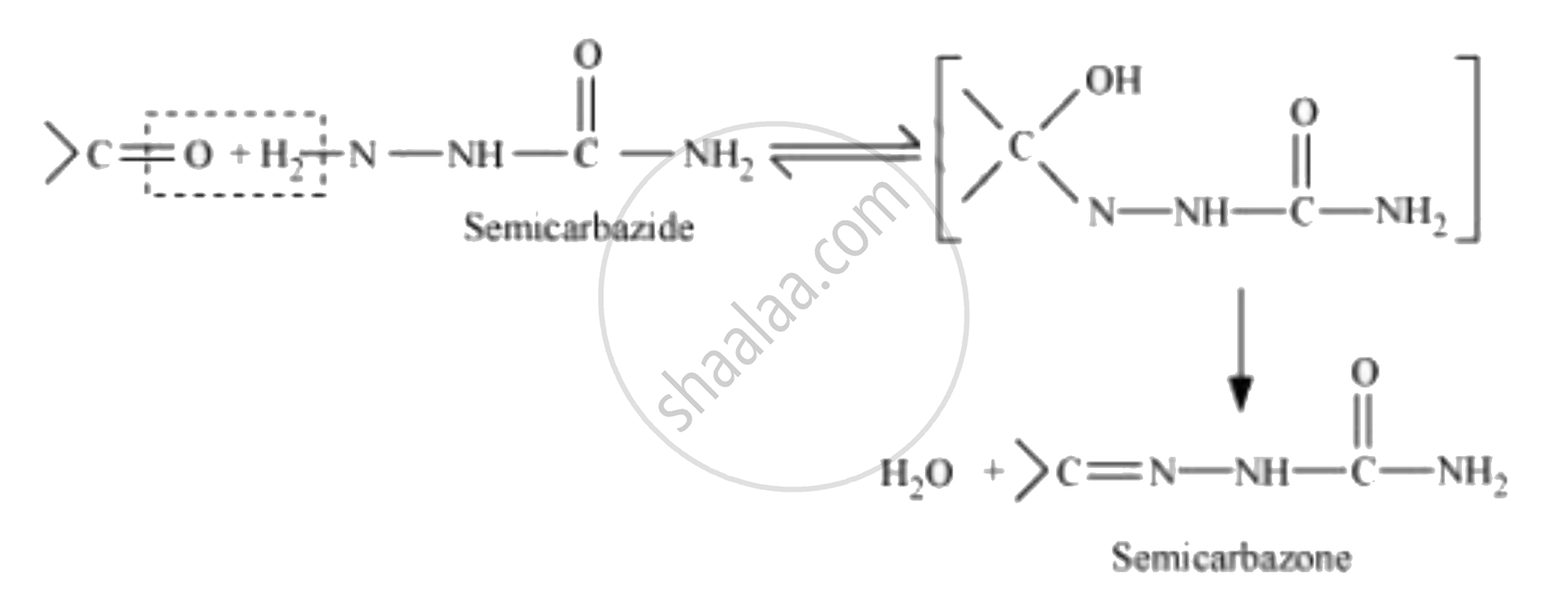
Semicarbazone
Advertisements
Solution
Semicarbazones are derivatives of aldehydes and ketones produced by the condensation reaction between a ketone or aldehyde and semicarbazide.
APPEARS IN
RELATED QUESTIONS
Arrange the following in the increasing order of their reactivity towards nucleophilic addition reaction:
C6H5COCH3, CH3-CHO, CH3COCH3
Predict the product of the following reaction:

Predict the product of the following reaction:
\[\begin{array}{cc}
\phantom{..............}\ce{O}\\
\phantom{..............}||\\
\ce{R - CH = CH - CHO + NH2 - C - NH - NH2 ->[H+]}\end{array}\]
Predict the products formed when cyclohexanecarbaldehyde reacts with the following reagents.
PhMgBr and then H3O+
How are the following compounds prepared?
benzaldehyde from benzoyl chloride
Write balanced chemical equations for action of ammonia on - acetaldehyde
Identify the compounds A, B and C in the following reaction.
\[\ce{CH3 - Br ->[Mg/ether] (A) ->[(i) CO][(ii) Water] (B) ->[CH3OH/H+][Δ] (C)}\]
Which one of the following gives only one monochloro derivative?
Which of the following has the most acidic hydrogen?
What is the action of sodium hypoiodite on acetone?
Aldehydes and ketones react with hydroxylamine to form ______.
Draw the structure of the following derivative.
The ethylene ketal of hexane-3-one
Draw structure of the following derivative.
The ethylene ketal of hexan-3-one
Draw structures of the following derivative.
The ethylene ketal of hexan-3-one
Draw the structure of the following derivative.
Acetaldehydedimethylacetal
Draw the structure of the following derivative.
The ethylene ketal of hexan-3-one
Draw structure of the following derivative.
The ethylene ketal of hexan-3-one
Draw the structure of the following derivative.
Acetaldehydedimethylacetal
Draw structure of the following derivative.
The ethylene ketal of hexan-3-one
